|
|
|
Copyright © 2004 by the author(s). Published here under license by The Resilience Alliance.
Go to the pdf version of this article.
The following is the established format for referencing this article:
Vo, M., D. Porter, G. Chandler, H. Kelsey, S. Walker, and B. E. Jones. 2004. Assessing photoinduced toxicity of polycyclic aromatic hydrocarbons in an urbanized estuary. Ecology and Society 9(5): 3. [online] URL: http://www.ecologyandsociety.org/vol9/iss5/art3/
A version of this article in which text, figures, tables, and appendices are separate files may be found by following this link.
Report, part of Special Feature on Urban Sprawl Assessing Photoinduced Toxicity of Polycyclic Aromatic Hydrocarbons in an Urbanized Estuary M. Vo1, D.E. Porter2, G.T. Chandler2, H. Kelsey2, S.P. Walker2, and B. E. Jones2
1South Carolina Department of Natural Resources; 2University of South Carolina
- Abstract
- Introduction
- Methods
- Results
- Discussion
- Conclusions
- Responses to this Article
- Acknowledgments
- Literature Cited
Increases in contaminants associated with urban sprawl are a particular concern in the rapidly developing coastal areas of the southeastern United States. Polycyclic aromatic hydrocarbons (PAHs) are contaminants associated with vehicle emissions and runoff from impervious surfaces. Increased vehicular traffic and more impervious surfaces lead to an increased loading of PAHs into coastal estuarine systems. The phototoxic effect of PAH-contaminated sediments on a sediment-dwelling meiobenthic copepod, Amphiascus tenuiremis, was estimated in Murrells Inlet, a small, high-salinity estuary with moderate urbanization located in Georgetown and Horry Counties, South Carolina, USA. Field-determined solar ultraviolet radiation (UV) and UV extinction coefficients were incorporated into laboratory toxicity experiments, and a model was developed to predict areas of specific hazard to A. tenuiremis in the estuary. The model incorporated laboratory toxicity data, UV extinction coefficients, and historical sediment chemistry and bathymetric data within a spatial model of sedimentary areas of the estuary. The model predicted that approximately 8–16% of the total creek habitat suitable for meiobenthic copepods is at risk to photoinduced PAH toxicity. This area is in the northern, more developed part of Murrells Inlet.
KEY WORDS: Hazard modeling, photoinduced toxicity, polycyclic aromatic hydrocarbons, spatial modeling, urbanized estuary.
Published: August 31, 2004
Urban sprawl and unplanned development are pervasive problems throughout the United States, causing rapid increases in population, roadways, and traffic as well as decreases in rural farmland and areas of undeveloped land (Mitchell 2001). Rapid population increases projected in the southeastern coastal United States (Allen and Lu 2000, Vernberg and Vernberg 2001) are of particular concern because of the related increases in development pressure on estuarine and coastal water resources. The populations of the U.S. coastal areas of South Carolina and Georgia are expected to increase by as much as 100% by 2026 (LU-CES 2000). These increases in population will lead to the development of more urbanization and sprawl. The associated increases in vehicular traffic and runoff from impervious surfaces such as paved roads are likely to both increase runoff flows and alter storm water composition, adding to the pressure on estuarine systems.
Estuaries are an economically valuable and highly productive coastal resource, and they serve many useful purposes (Vernberg et al. 1992, Rathbun 1998, Porter et al. 1996). These ecosystems have obvious aesthetic value, but they also provide habitat for commercially and ecologically significant shellfish, fish, and crustacean species (Rathbun 1998). Nearshore and estuarine fish comprise 50% of the world’s harvest (Kennish 1992), and the majority of these species depend on meiobenthic copepods for food during the early stages of life (Coull 1990). As coastal areas become increasingly populated, the ecological function and integrity of estuarine areas are threatened.
More than 50% of the population of the United States is estimated to live and work in coastal counties that comprise only 10% of the country’s landmass (Vernberg et al. 1992). As a result of this anthropogenic pressure, estuarine areas over the past century have served as a repository for a variety of industrial and municipal wastes, dredged materials, and sewage sludge. These loadings have led to widespread contamination by heavy metals, pesticides, petroleum hydrocarbons, nutrients, and pathogens (Kennish 1992). Understanding and monitoring the impacts of contaminant loading may help managers make decisions to preserve the ecological function of coastal areas (Kennish 1992, Vernberg et al. 1992, Rathbun 1998).
One widespread class of urban contaminants is polycyclic aromatic hydrocarbons (PAHs). These are generated mainly during the combustion of fossil fuels (Neff 1979, Huang et al. 1993). PAHs bind readily to particulate surfaces and are relatively stable in their ground states. These contaminants are persistent in substrates such as marsh sediment, particularly when the sediment is characterized by low oxygen and high organic carbon contents (Arfsten et al. 1996).
Traditionally, testing the toxicity of PAH-contaminated sediments has been conducted in the absence of UV to avoid photodegradation of intact PAHs. The result is an overall underestimation of PAH toxicity in environmental risk assessments (Arfsten et al. 1996). When modifying water quality criteria, the U.S. Environmental Protection Agency (EPA) recognized that photoinduced PAH toxicity poses an additional risk to aquatic organisms, yet the modification of current sediment quality criteria has not received comparable attention (Ankley et al. 1995). Benthic organisms may be at greatest risk for the phototoxic effects of PAH-contaminated sediments because their ecology places them in close association with these sediments (Monson et al. 1995). Few studies have examined the phototoxic effects of PAH-contaminated sediments on benthic organisms.
Research has shown that photodegradation of PAHs is directly proportional to the intensity of incident solar radiation (Wortham et al. 1993). Literathy et al. (1990) investigated the fluorescence of PAHs in Kuwaiti crude oil with respect to seawater absorption, suspended sediments, and solar radiation levels. Fluoresence is used to characterize the photolysis of PAHs. The authors found that the photolysis of PAHs may occur prior to the photoexcitation and subsequent photoenhanced toxic activities of these contaminants. They showed that, in August, maximum PAH photolysis occurred at midday with radiation wavelengths of 330–350 nm in the UV-A range, with only a 6% decrease in photolysis at a depth of 1 m. In marine waters, the rate of photochemical reactions involving PAHs is governed by many factors, including UV radiation intensity and the specific absorption wavelengths for individual PAHs (Literathy et al. 1990).
Because of the projected population increases around Murrells Inlet, concurrent increases in the amount of area developed, the expansion of impervious surfaces, and greater vehicular traffic, PAH contamination in the Murrells Inlet estuary is expected to increase. This study evaluates an ecological hazard associated with the PAH contamination that results from increased development. The specific objective of this study is to identify geographic areas within the Murrells Inlet estuary that could be considered hazardous to A. tenuiremis because of photoinduced toxicity in PAHs. Hazard criteria were created based upon laboratory phototoxicity bioassays, habitat considerations, environmental solar UV intensity, and PAH contamination levels.
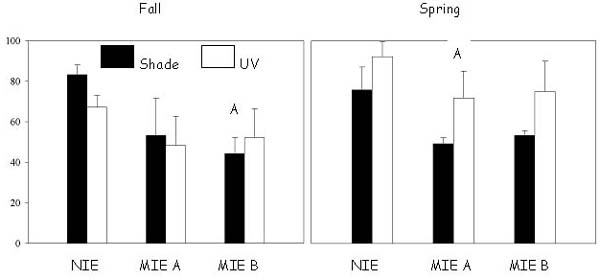
Murrells Inlet estuary (MIE) is a high-salinity, 26.7 km2 estuary containing urbanized uplands, marshland, and water. It is located approximately 122 km northeast of Charleston, South Carolina, USA (Fig. 1). Murrells Inlet is characterized by semidiurnal tides with a mean range of 1.37 m. The area has been experiencing high rates of residential and commercial development to satisfy the demands of a growing coastal population (Porter et al. 1997).
|
Fig. 1. Murrells Inlet land use.
|
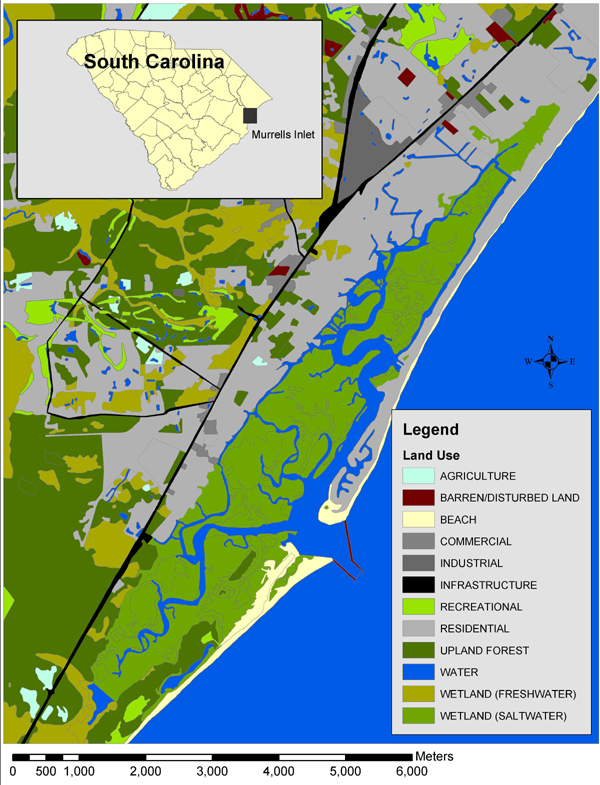
Although PAH levels vary throughout Murrells Inlet, they are highest in locations adjacent to developed areas, such as marinas and highway overpasses (USES Annual Report 1994; Fig. 2). PAH concentrations were measured in Murrells Inlet by Fortner et al. (1996). The mean total PAH concentration in Murrells Inlet is 518 ng/g. The PAHs measured were phenanthrene, anthracene, fluoranthene, pyrene, benzo(a)anthracene, chrysene, benzo(b)fluoranthene, benzo(k)fluoranthene, benzo(a)pyrene, and benzo(ghi)perylene. These PAHs excite in the UV-B spectrum and emit in the UV-A and visible spectra (Futoma 1981).
|
Fig. 2. Estimates of levels of polycyclic aromatic hydrocarbons (PAHs) in sediments from Murrells Inlet.
|
North Inlet estuary (NIE) was used as the control site in toxicity tests. North Inlet is located 70 km northeast of Charleston, South Carolina, USA, and approximately 32 km south of Murrells Inlet (Fig. 1). It comprises 80 km2of undeveloped barrier islands, marshland, and upland forest, and it exhibits consistently low total PAH concentrations (Fortner et al. 1996; Table 1).
|
Table 1. Summary of individual polycyclic aromatic hydrocarbons (PAH) in sediments from two Murrells Inlet Estuary sites (MIE A and MIE B) for the October 1998 (Fall) and May 1999 (Spring) toxicity tests (ng/g dry weight). North Inlet Estuary (NIE) control site data is from Fortner et al. 1996.
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
A. tenuiremis is an amphi-Atlantic, sediment-ingesting, benthic, harpacticoid copepod that has been used successfully in several field and laboratory-spiked sediment toxicity studies (Chandler and Green 1996, Wirth et al. 1998). It is an easily cultured, sexually dimorphic, infaunal copepod with holobenthic larvae. Its offspring occur in easily distinguishable clutches and juvenile stages for post-test, life-stage enumeration (Chandler and Green 1996).
Sediment PAH analysesTriplicate sediment samples from the NIE control site and from two Murrells Inlet sites (MIE A and MIE B; Fig. 2) were analyzed for PAHs at the National Oceanic and Atmospheric Administration Center for Coastal Environmental Health and Biomolecular Research Laboratory in Charleston, South Carolina, USA (Fortner et al. 1996). The procedure for measuring organic contaminants in sediments was modified from Krahn et al. (1988), Sanders (1995), and Schantz et al. (1997). The following PAHs were tested: anthracene, fluoranthene, pyrene, benzo(a)anthracene, chrysene, benzo(b)-fluoranthene, benzo(k)fluoranthene, benzo(a)pyrene, benzo(g,h,i)perylene, naphthalene, 1-methylnaphthalene, 2-methylnaphthalene, biphenyl 2,6 dimethylnaphthalene, acenaphthene, acenaphthylene, 2,3,5 trimethylnaphthalene, fluorene, 1-methylphenanthrene, perylene, ideno(1,2,3-cd)pyrene, and dibenz(a,h)anthracene (Table 1).
Phototoxicity test designLaboratory reproduction bioassays were used to determine whether toxicity to A. tenuiremis was greater with co-exposure to PAH-contaminated sediments from Murrells Inlet and simulated UV compared to the same sediments that have not been irradiated with UV, i.e., UV-shaded. Copepod chronic reproductive bioassays were conducted at known UV intensities and four site-specific concentrations of sediment contaminants. These data were combined with field UV intensity data to determine whether the UV levels measured in the laboratory were consistent with those in the natural habitat of our organism. The phototoxicity test design was modified from the benthic copepod sediment toxicity test developed by Chandler and Green (1996).
NIE control and MIE contaminated sediments were press-sieved on 63 μm stainless steel sieves prior to testing to remove large particulates and meiofauna. Sediment tests were replicated nine times: tests of UV-exposed contaminated sediments were replicated five times, and tests of UV-shaded contaminated sediments were replicated four times. All test glassware was acid-washed and rinsed with 1:1 acetone-hexane to neutralize glass surfaces and minimize adsorption of sediment PAHs to the glass substrate. Test chambers were made from 50 mL glass beakers with 2.5 cm holes blown in them. The holes were covered with 63 μm mesh to allow seawater to flow through the test chambers. For each sediment type, 10 mL of sediment was covered with 25 mL of filtered, artificial seawater (ASW), and 30 gravid A. tenuiremis per replicate were harvested from stock sediment cultures. These were loaded into the 50 mL glass beakers.
A total of 30 individual test beakers were placed under constant ASW flow in an environmental chamber held constant at 30 ± 1 ppt salinity, 20 ± 1°C, and 7.5 ± 0.1 pH. Two 1 m long, 40-watt, 340-nm UV bulbs (Q-Panel Lab Products, Cleveland, Ohio) were placed above test chambers as a UV source on a 16:8 h light:dark cycle (L:D). Full-spectrum fluorescent light bulbs were placed 15 cm below the test chambers to provide a non-UV source on a 16:8 h L:D. UV intensity was measured every three days. Each beaker received 21 x 106 cells/mL phytoplankton food every three days. An IL1400-3987 photometer along with submersible SUL-005 and SED-240 detectors (International Light Inc., Newburyport, Massachusetts, USA) measured UV-A and UV-B, respectively. Non-UV control shields were made of 500 mL amber polypropylene bottles with the bottom 2 cm removed so that they could be placed over the test beakers to block the passage of UV to the UV-shaded control chambers.
UV extinction in seawater was measured prior to each toxicity test to determine the UV-A and UV-B exposures at the sediment-water interface. Extinction coefficients for UV-A and UV-B were derived using a 2 L rectangular container filled with ASW from the flow-through system used in the toxicity tests. The container was placed beneath the UV bulbs in the location later occupied by the toxicity test setup. UV-A and UV-B were measured at the air-water interface and at 1 cm increments down to the container bottom. Extinction coefficients for UV-A and UV-B were calculated for the 1 cm depth (the average depth to the sediment-water interface) using the following formula (Kirk 1994):
|
Id = Ioe-Ke(d)
| (1) |
where Ke = UV extinction coefficient,
d = depth,
Id = UV intensity at depth,
and Io = initial UV intensity.
A. tenuiremis was cultured for 14 d in these sediments while exposed to UV or not exposed to UV, i.e., UV-shaded. At test end, surviving copepods in the gravid, naupliar, copepedite, and adult life stages were sieved onto a 53 μm sieve, checked for dead individuals, preserved in 5% formalin and rose bengal, and enumerated for sex, life stage, and clutch size.
Statistical analysesAll toxicity test data were analyzed using Microsoft Excel, SAS, and MINITAB software. Standard assumptions of normality and independence of observations were evaluated and found to be valid. Multiple regression techniques were performed on data from replicate experiments to determine which of the following independent variables were significant predictors of toxicity end points (P > 0.95): season, presence/absence of UV shade, sediment from the contaminated (MIE) or control (NIE) sites, and their interaction. Once predictors for end points were established, estimates of means for UV and UV-shaded treatments were compared using ANOVA in SAS. SAS LSMEANS was used for pairwise comparisons among UV-exposed and UV-shaded treatments (α = 0.05). This resulted in an actual individual comparison significance value of 0.006 based on nine predetermined comparisons. Toxic end points included adult mortality, percentage of females gravid at test end, mean clutch size, and realized offspring production. Based on Chandler and Green (1996), production was determined by the following formula:
|
offspring production = (copepidites + nauplii)/surviving females at 14 d
| (2) |
To qualify as a hazard area for A. tenuiremis in Murrells Inlet, locations were required to meet three criteria: (1) they needed to have PAH levels equal to the minimum observed to cause toxicity to A. tenuiremis in the laboratory, (2) they had to have UV levels equal to the minimum observed to cause toxicity to A. tenuiremis in the laboratory, and (3) they had to be designated as habitat for A. tenuiremis.
Areas of Murrells Inlet that had PAH and UV levels above the minimum observed to be toxic to A. tenuiremis in the laboratory were identified using continuous surface estimates of PAH and UV levels. These surface estimates were derived using various GIS techniques and statistical analyses, including kriging, from PAH measurements taken from 30 sample sites (Fortner et al. 1996) and field-measured UV levels in the estuary.
Site-specific total PAH values, extinction coefficients, and monthly average surface UV intensity values for each of the 30 sample sites along with corresponding latitudinal and longitudinal coordinates were entered into the GS+ geostatistical software (Gamma Design Software, Plainwell, Michigan, USA). Semivariance analyses were conducted before proceeding with spatial interpolation of each parameter. The kriging option in the GS+ package was used to develop continuous surfaces of UV at the air-water interface, UV extinction coefficients, and total PAH levels in sediment. After kriging, each data layer was imported into Arcview (Environmental Systems Research Institute, Redlands, California, USA). Maps of all continuous surface data were generated and converted to grids that divided spatial attributes into discrete numeric units. These grids were used in algebraic algorithms in ARC/GRID (Environmental Systems Research Institute, Redlands, California, USA).
A bathymetric model of Murrells Inlet was generated using a differential global positional system and a depth finder to obtain real-time depth measurements. The result is an extensive spatial model of mean low-water depth, i.e., average depth at low tide, throughout the estuary. Summary statistics were obtained using the MINITAB statistical package. Measurements of UV intensity from band A, i.e., wavelengths of 320–400 nm, were obtained during concurrences of low tide and 12:00 noon ± 2 h in an effort to characterize potential high-hazard, midday UV conditions. The UV-B band, i.e., wavelengths of 280–320 nm, was not measured in the field because of tide and time limitations that precluded the measurement of both bands of UV. Measurements were obtained with the SUL-005 detector at the sediment-water interface along a shore-to-depth transect to a depth at which UV was approximately 1% of that measured at the surface (UV-A ≈ 25 μW/cm2). UV-A extinction coefficients were determined from plots of light intensity vs. depth (Kirk 1994). Ten sites were measured monthly, and all 30 sample sites were measured during October 1998, May 1999, and August 1999. UV extinction coefficients (Ke) for each site and month were determined from natural logs of linear plots of UV-A values vs. depth (Kirk 1994):
|
Ke = 1n(Io/Id)/z
| (3) |
where Ke = UV extinction coefficient,
z = depth,
Io = UV intensity at surface,
and Id = UV intensity at depth.
U.S. National Wetlands Inventory (NWI) data for Murrells Inlet were used to identify areas that were classified as appropriate intertidal and subtidal habitat for A. tenuiremis. These areas extended from the tidal creek fringe dominated by Spartina alterniflora (smooth cordgrass) to the low intertidal mudflat.
To generate the final hazard model, cells in the PAH hazard grid, the UV hazard grid, and the habitat grids were assigned values and multiplied across grids. UV hazard grid cells for each month were assigned a value of 2 if their UV levels were equal to or greater than the minimum level toxic to A. tenuiremis at 1 cm depth in the laboratory. All other cells were assigned a value of 1. Similarly, PAH hazard grid cells with PAH levels equal to or greater than the minimum laboratory level to contribute to photoenhanced toxicity for A. tenuiremis were assigned a value of 2. Each UV hazard grid was multiplied by the PAH hazard grid, resulting in a composite grid of hazard for the estuary. Hazard grids were then multiplied by the suitable habitat grid. The habitat grid cells were coded 2 if the NWI classification corresponded to one of the following habitat types: Estuarine Subtidal Unconsolidated Bottom, Estuarine Intertidal Unconsolidated Shore, Regularly Flooded, Estuarine Subtidal Unconsolidated Bottom, or Estuarine Intertidal Unconsolidated Shore, Irregularly Flooded. The resulting grid represents areas that meet all three criteria noted above as potentially hazardous to A. tenuiremis in Murrells Inlet.
Key results for copepod survival, UV intensity, and hazard modeling are presented below; supplemental results, discussion, and conclusions are shown in Appendix 1.
Copepod survivalIn the October 1998 experiment, no significant UV-related mortality was observed for adult females in sediments from the contaminated sites at the Murrells Inlet estuary (MIE) or the control sites at the North Inlet estuary (NIE). However, in the October 1998 toxicity test, the UV-shaded treatment with the control sediments yielded significantly higher survival than UV-shaded treatment of contaminated sediments (P = 0.0009). During the May 1999 experiment, copepod adult female survival was significantly higher in UV-exposed sediments from the control site.
Survival was significantly lower in UV-exposed contaminated sediments from MIE A than in UV-exposed sediments from the control site (P = 0.0035). The presence of UV, sediment contamination, and season were all significant predictors of survival in both experiments (ANOVA; P < 0.05). A summary of copepod survival is shown in Fig. 3.
UV intensity
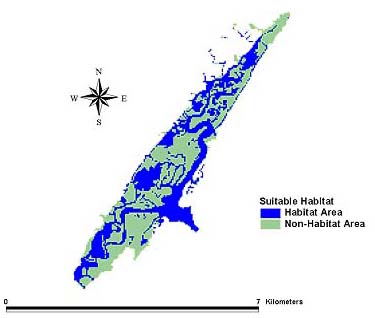
Suitable Amphiascus tenuiremis habitat in Murrells Inlet was mapped using the U.S. National Wetlands Inventory data (Fig. 4). UV intensities for the months of October 1998, May 1999, and August 1999 were developed for A. tenuiremis habitat on the bottom, i.e., the sediment-water interface. In May 1999, the maximum derived UV intensity at the sediment-water interface was 2344 (μW/cm2, and 63% of total suitable organism habitat in the estuary showed UV intensities at the bottom equal to or greater than the minimum observed to cause toxicity to A. tenuiremis in the laboratory. In August 1999, the maximum UV intensity at the bottom was 2143 μW/cm2, with 63% of suitable habitat areas meeting or exceeding the toxicity minimum in the laboratory. In October 1998, the maximum bottom UV was 1635 μW/cm2, and 62% of areas in the creek beds showed UV intensities equal to or greater than the laboratory-determined UV minimum. Seasonal results are shown in Figs. 5–7.
|
Fig. 4. Suitable habitat for Amphiascus tenuiremis based on the U.S. National Wetlands Inventory classification of Murrells Inlet.
|
|
Fig. 5. Intensity of ultraviolet light in the UV-A range (μW/cm2) on the bottom (the sediment-water interface) during low tide for October 1998.
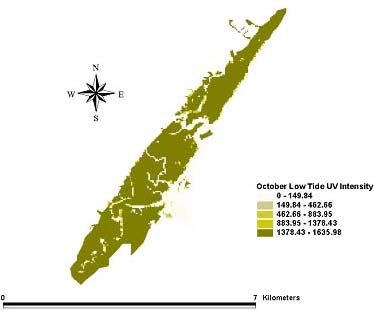
|
|
Fig. 6. Intensity of ultraviolet light in the UV-A range (µW/cm2) at the bottom (the sediment-water interface) during low tide for May 1999.
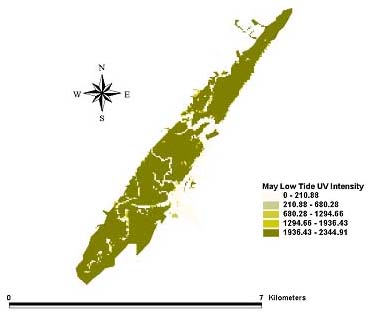
|
|
Fig. 7. Intensity of ultraviolet light in the UV-A range (μW/cm2) at the bottom (the sediment-water interface) during low tide for August 1999.
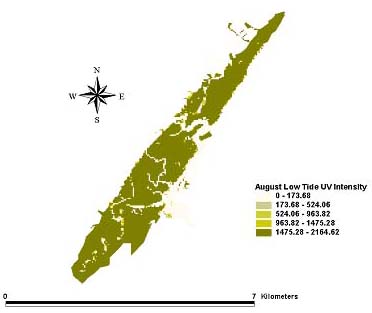
|
Distribution patterns of UV intensity on the bottom were found to be similar for all three months. Deeper areas near the mouth of the inlet were characterized by little or no UV penetration. Shallower parts of the northern and southern inland areas saw the highest UV penetration at the bottom. The inland areas of the extreme northern and southern portions of the estuary received enough UV at the bottom to reach the laboratory minimum for causing toxicity to A. tenuiremis. This suggests that the northern, urbanized portion of the estuary creek bed, with its high total PAH levels, could be susceptible to photoenhanced toxicity. For some parts of the inland estuarine areas, depths were 0 m at low tide, and UV on the creek bed was equal the initial UV intensity. UV intensity on the bottom appeared consistent across the months of May, August, and October despite seasonal trends in UV extinction levels. This may be because trends in UV levels at the surface may counteract seasonal trends in extinction.
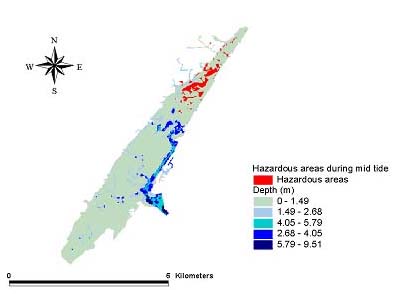
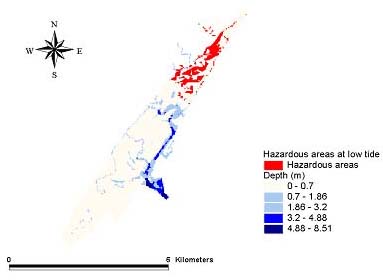
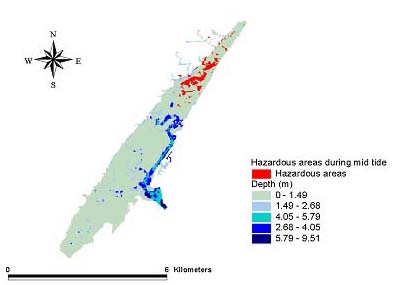
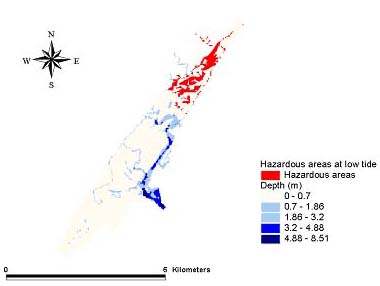
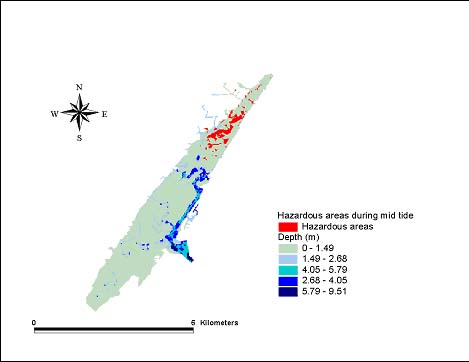
Hazard modelsGeographic areas that could be considered hazardous because of phototoxicity to A. tenuiremis within the Murrells Inlet estuary were mapped for low and mid tide during the months of October 1998, May 1999, and August 1999 (Figs. 8–13). For all three months, these hazardous areas were located during low tide in the urbanized extreme northern portion of the estuary, mainly on the inland side. In May 1999, 929,416 m2 or approximately 16% of the suitable A. tenuiremis habitat was susceptible to phototoxic effects during low tide. In the creek bed, susceptible areas were either exposed during low tide or less than 0.5 m deep. For August 1999, also during low tide, 16% (935,072 m2) of suitable habitat was susceptible. In October 1998, 16% of suitable habitat was susceptible (929,416 m2) at low tide. At mid tide, the areas susceptible to phototoxic effect were reduced by half for all three months. Approximately 503,355 m2 or 8.5% of suitable habitat was susceptible in May 1999, 8.7% or 509,011 m2 in August 1999, and 8.4% (495,814 m2 ) in October 1998. These areas were also in the northern urbanized portion of the estuary. No areas were susceptible as a phototoxicity hazard to A. tenuiremis during high tide.
|
Fig. 8. The October 1998 low tide hazard for Amphiascus tenuiremis from photoinduced toxicity of polycyclic aromatic hydrocarbons.
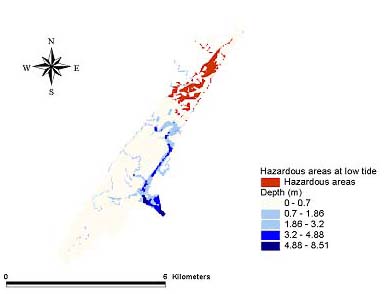
|
|
Fig. 9. The October 1998 mid tide hazard for Amphiascus tenuiremis from photoinduced toxicity of polycyclic aromatic hydrocarbons.
|
|
Fig. 10. The May 1999 low tide hazard for Amphiascus tenuiremis from photoinduced toxicity of polycyclic aromatic hydrocarbons.
|
|
Fig. 11. The May 1999 mid tide hazard for Amphiascus tenuiremis from photoinduced toxicity of polycyclic aromatic hydrocarbons.
|
|
Fig. 12. The August 1999 low tide hazard for Amphiascus tenuiremis from photoinduced toxicity of polycyclic aromatic hydrocarbons.
|
|
Fig. 13. The August 1999 mid tide hazard for Amphiascus tenuiremis from photoinduced toxicity of polycyclic aromatic hydrocarbons.
|
Creek beds considered hazardous to A. tenuiremis because of phototoxicity during low and mid tide were located in the northern portions of the estuary in all three months. The amount of area susceptible to photoxic effects during mid tide was significantly reduced relative to those at low tide. These results suggest that depth has implications for our hazard model. Most of the area considered susceptible at low tide for all months was less than 0.32 m deep, and the total area did not change substantially during these months. This is contrary to what was expected, because neither surface UV intensity nor UV extinction was consistent throughout the year. Surface UV intensity was highest during the summer months, when UV extinction levels were also high. The converse was true of the winter months. Peaks in UV extinction during the summer could be caused by sediment resuspension and phytoplankton population dynamics (see above). The result of these seasonal trends is a counteraction that causes little or no seasonal change in the UV intensity reaching the bottom where A. tenuiremis lives.
The area in which phototoxicity poses the greatest hazard to A. tenuiremis in the northern portion of Murrells Inlet is characterized by more residential and industrial development than other parts of the inlet. All susceptible areas were within 600 m of urbanized sections, suggesting that urbanization may be linked to the potential for photoenhanced toxicity. These areas also had the highest levels of total polycyclic aromatic hydrocarbons (PAHs). Initially, these areas were expected to be hazardous because of their PAH content, but high extinction levels were ultimately expected to reduce if not eliminate this hazard. This expectation was strengthened by the findings of Skjemstad et al. (1993), who showed that PAHs associated with sediments high in silt clay content were protected from photo-oxidation by UV. The results of the present study support these findings. These areas are only likely to be susceptible to photoenhanced toxicity when the water is very shallow, allowing UV to penetrate to the PAH-contaminated bottom sediments. Over all, the hazard to A. tenuiremis from the photoenhanced toxicity of Murrells Inlet sediments exists for only approximately 16% of the habitat suitable for the copepod, regardless of the season. Approximately 8% of the estuary is considered hazardous to A. tenuiremis during mid tide.
These results clearly show that development-related contaminants have an impact on the ecology of an urbanized estuary. Within the Murrells Inlet estuary (MIE), the areas most susceptible to enhanced phototoxicity from polycyclic aromatic hydrocarbons (PAHs) were located in the northern, urbanized portions of the estuary. Because PAHs result from fossil fuel combustion, the appearance of these chemicals in Murrells Inlet is assumed to be related to storm water drainage. Increases in PAH contamination from storm water runoff are expected to continue in the Murrells Inlet estuary and other estuaries in the southeastern United States as development pressure expands areas of urbanization and traffic. Increases in PAH contamination, in turn, will lead to increased toxicity to key organisms. Previous work on benthic macrofauna suggests that toxicity in UV-exposed sediment in the Murrells Inlet estuary is higher than toxicity in North Inlet sediment (control). The highest toxicity was observed in the sediments characterized by highest PAH levels. Experiments conducted without UV indicate that PAH-contaminated sediments from Murrells Inlet were more toxic to A. tenuiremis than North Inlet sediments (Kovatch et al. 1999, Wirth et al.
1998). Results from the present study suggest that toxicity of PAH-contaminated sediments from Murrells Inlet is enhanced with co-exposure to UV. In the October 1998 experiments, realized offspring production and clutch size were
both affected by PAH-contaminated MIE sediments with UV exposure relative to the same sediments without UV exposure. Interestingly, the phototoxic response did not vary between MIE sites despite differences in the amount of PAH contamination. This suggests that there is no relationship between the amount of PAH and the phototoxicity in these
sediments. One explanation for this may be that, although PAH is more toxic in the presence of UV, the phototoxic response measured in these experiments was caused by the presence of another contaminant in these field-collected sediments (see Ankley 1994). Another possibility is that differences in PAH-related phototoxicity cannot be measured at these PAH levels. In this study, co-exposure of meiobenthic copepods to UV and PAH-contaminated sediments resulted in increased mortality, enhanced reproductive toxicity, and reduced clutch sizes. However, toxic responses of A. tenuiremis to PAH and UV co-exposure were end point- and season-specific. The fact that season was a significant variable in all the regression models provides evidence that seasonal differences in sediment toxicity can be important. These differences are likely manifest as changes in the biogeochemical properties of the sediments and in the relative composition of sediment mixtures, e.g., labile and refractory organic carbon, grain size, etc. Additional experiments to identify the toxic effects of individual PAHs should include sediments spiked in a manner that promotes the identification and evaluation of additive toxicity to mimic the predominant PAHs in this estuary. See Appendix 1 for continued discussion. This study provides some insight into the potential ecological hazards associated with PAH contamination resulting from increased development pressure. Population increases accompanied by more development, impervious
surfaces, and vehicular traffic, i.e., sprawl, in the Murrell’s Inlet area suggest that PAH contamination in the Murrells Inlet estuary is likely to increase as well. The aesthetic, economic, and ecologic value of estuaries demands that
the impacts of these kinds of anthropogenic activities be more precisely evaluated before the function and integrity of estuarine systems are further compromised.
Integration is the key to a successful environmental impact assessment and systems analysis (Fedra 1994). Managers should therefore make use of available computer technologies that enhance the integration process (O’Regan 1996) and provide visually oriented environmental models to support their decisions (Giertsen 1994). In the present study, laboratory data were integrated with field data and historical data on polycyclic aromatic hydrocarbons (PAHs) from Murrells Inlet to establish a spatial model of the potential hazard from the photoenhanced toxicity of PAHs in Murrells Inlet estuary, even if the small number of site samples processed in laboratory bioassays limits the comprehensiveness of these hazard maps. This study may provide general information about the minimum level of PAH contamination to induce a phototoxic response from A. tenuiremis.
Based upon laboratory observations of toxicity at specific UV levels and PAH concentrations and historical site-specific PAH level data, predictions were made of the potential for photoinduced toxicity at locations in the estuary
that are characterized by levels similar to or greater than those levels tested in the laboratory. Predictions at specific sites not tested in the laboratory were also made. These further predictions were based upon historical data on
the distributions and concentrations of PAHs throughout the Murrells Inlet estuary. The end result is a series of maps that represent the hazard presented by photoinduced toxicity during three different seasons of the year. See
Appendix 1 for additional conclusions. Research that integrates laboratory and spatial modeling techniques can provide important descriptive data for setting sediment-quality criteria and for developing science-based coastal regulations and monitoring. In this case,
hazards to A. tenuiremis associated with increased PAH contamination in an urbanized estuary were demonstrated through the integration of laboratory and spatial modeling techniques. Although the model was conservative, the effects of development-related PAH toxicity on a key species, A. tenuiremis, were clear. The Murrells Inlet estuary is typical of other developing or urbanized estuaries in the southeastern United States. Other estuaries experiencing high growth and the resultant increases in impervious surfaces and vehicular traffic are likely to exhibit similar effects. Responses to this article are invited. If accepted for publication, your
response will be hyperlinked to the article. To submit a comment, follow this
link. To read comments already accepted, follow this link.
Responses to this article are invited. If accepted for publication, your response will be hyperlinked to the article. To submit a comment, follow this link. To read comments already accepted, follow this link.
Acknowledgments
The National Ocean Service (NOS) and the University of South Carolina (USC) do not approve, recommend, or endorse any proprietary
product or material mentioned in this publication. No reference shall be made
to NOS or USC or to this publication furnished by NOS or USC in any advertising
or sales promotion that would indicate or imply that NOS or USC approves,
recommends, or endorses any proprietary product or proprietary material
mentioned herein or that has as its purpose any intent to cause directly or
indirectly the advertised product to be used or purchased because of NOS or USC
publication. This publication represents manuscript number 1345 of the Baruch Marine
Science Library. The research described within was supported, in part, by a grant from the
National Oceanic and Atmospheric Administration (#NA06OP0358) awarded to the
University of South Carolina. The authors also gratefully acknowledge the contributions of David White,
Matt Neet, and William (Jeff) Jefferson of the Baruch Institute.
Allen, J., and K.S. Lu. 2003. Modeling and prediction of future urban growth in the Charleston region of South Carolina. Conservation Ecology 8: 2. [online] URL:http://www.consecol.org/vol8/iss2/art2/.
Ankley, G. T., S.A. Collyard, P.D. Monson, and P.A. Kosian. 1994. Influence of ultraviolet light on the toxicity of sediments contaminated with polycyclic aromatic hydrocarbons. Environmental Toxicology and Chemistry 13: 1791-1796.
Ankley, G. T., R.J. Erickson, G.L. Phipps, V.R. Mattson, P.A. Kosian, B.R. Sheedy, and J.S. Cox. 1995. Effects of light intensity on the phototoxicity of fluoranthene to a benthic macroinvertebrate. Environmental Science and Technology 29: 2828-2833.
American Society for Testing and Materials. 1980. Standard practices for conducting toxicity tests with fishes, macroinvertebrates, and amphibians. Annual book of ASTM standards. ASTM, Philadelphia, USA.
Arfsten, D. P., D.J. Schaeffer, and D.C. Mulveny. 1996. The effects of near ultraviolet radiation on the toxic effects of polycyclic aromatic hydrocarbons in animals and plants: a review. Ecotoxicology and Environmental Safety 33: 1-24.
Boese, B. L., J.O. Lamberson, R.C. Swartz, R.J. Ozretich, and F. Cole. 1998. Photo-induced toxicity of PAHs and alkylated PAHs to a marine infaunal amphipod (Rhepoxynius abronius). Archives of Environmental Contamination and Toxicology 34: 235-240.
Carpenter, E.J. 1971. Annual phytoplankton cycle of the Cape Fear River Estuary, North Carolina. Chesapeake Science 12: 95-104.
Chandler, G.T., and A.S. Green. 1996. A 14-day harpacticoid copepod reproduction bioassay for laboratory and field contaminated muddy sediments. New Techniques in Aquatic Toxicology Ch. 3(NA): 23-39.
Coull, B.C. 1990. Are members of meiofauna food for higher trophic levels? Transactions of the American Microscopical Society 109: 233-246.
Fedra, K. 1994. Integrated environmental information and decision support systems. Pages 269-288 in G. Guariso and B. Page, editors. Computer support for environmental impact assessment. Elsevier Science, B.V. North-Holland, Amsterdam, Netherlands.
Fortner, A.R., M. Sanders, and S.W. Lemire. 1997. Polynuclear aromatic hydrocarbon and trace metal burdens in sediment and the oyster, Crassostrea virginica (Gmelin), from two high-salinity estuaries in South Carolina. Pages 445-477 in F.J. Vernberg, W.B. Vernberg, and T. Siewicki, editors, Sustainable development in the southeastern coastal zone. University of South Carolina Press, Columbia, USA.
Futoma, D.J., S.R. Smith, T.E. Smith, and J. Tanaka. 1981. Polycyclic aromatic hydrocarbons in water systems. CRC Press, Boca Raton, Florida, USA.
Giertsen, C., and A. Lucas. 1994. 3d visualization for 2D GIS: an analysis of the users’ needs and a review of techniques. Eurographics 13: C1-C12.
Holst, L.L., and J.P. Giesy. 1989. Chronic effects of photo-enhanced toxicity of anthracene on Daphnia magna reproduction. Environmental Toxicology and Chemistry 8: 933-942.
Huang, X., D.G. Dixon, and B.M. Greenberg. 1993. Impacts of UV radiation and photomodification on the toxicity of PAHs to the higher plant Lemna gibba (duckweed). Environmental Toxicology and Chemistry 12: 1067-1077.
Ireland, D. S., G.A. Burton, Jr., and G.G. Hess. 1996. In situ toxicity evaluations of turbidity and photoinduction of polycyclic aromatic hydrocarbons. Environmental Toxicology and Chemistry 15: 574-581.
Kennish, M.J. 1992. Ecology of estuaries: anthropogenic effects. CRC Press, Boca Raton, Florida, USA.
Kirk, J.T.O. 1994. Light and photosynthesis in aquatic ecosystems. Cambridge University Press, New York, USA.
Kovatch, C.E., G.T. Chandler, and B.C. Coull. 1999. Utility of a full life-cycle copepod bioassay approach for assessment of sediment-associated contaminant mixtures. Marine Pollution Bulletin 38: 692-701.
Krahn, M.M., C.A. Wigren, R.W. Pearce, L.K. Moore, R.G. Boger, W.D. McLeod, Jr., S.L. Chan, and D.W. Brown. 1988. New HPLC cleanup and revised extraction procedures for organic contaminants. National Oceanic and Atmospheric Administration (NOAA) Technical Memorandum NMFS F/NWC-153: 23-47.
Literathy, P., G. Morel, and A. Al-Bloushi. 1990. Environmental transformation, photolysis of fluorescing petroleum compounds in marine waters. Water Science and Technology 23: 507-516.
LU-CES (Land Use — Coastal Ecosystem Study). 2000. Overview Report. Retrieved November 5, 2002, from http://www.lu-ces.org/overview.htm.
Mallin, M.A., H.W. Paerl, and J. Rudek. 1991. Seasonal phytoplankton composition, productivity and biomass in the Neuse River Estuary, North Carolina. Estuarine. Coastal and Shelf Science 32: 609-623.
Mitchell, J.G. 2001. Urban sprawl: the American dream? National Geographic 200(July, 2001): 48-73
Monson, P. D., G.T. Ankley, and P.A. Kosian 1995. Phototoxic response of lumbriculus variegatus to sediments contaminated by polycyclic aromatic hydrocarbons. Environmental Toxicology and Chemistry 14: 891-894.
Neff, J.M. 1979. Polycyclic aromatic hydrocarbons in the aquatic environment. Sources, fate, and biological effects. Applied Sciences, London, UK.
Newsted, J.L., and J.P. Giesy. 1987. Predictive models of photo-induced acute toxicity of polycylic aromatic hydrocarbons to Daphnia magna, Strauss (Cladocera, Crustacea). Environmental Toxicology and Chemistry 6: 445-461.
O’Regan, P.R. 1996. The use of contemporary information technologies for coastal research and management—a review. Journal of Coastal Research 12: 192-204.
Patten, B.C., R.A. Mulford, and J.E. Warinner. 1963. An annual phytoplankton cycle in the Lower Chesapeake Bay. Chesapeake Science 4: 1-20.
Porter, D.E., W.K. Michener, T. Siewicki, D. Edwards, and C. Corbett. 1997. Utilizing the tools of geographic information processing to assess the impacts of urbanization on a localized coastal estuary: a multi-disciplinary approach. Pages 355-388 in F.J. Vernberg, W.B. Vernberg, and T. Siewicki, editors, Sustainable development in the southeastern coastal zone. University of South Carolina Press, Columbia, USA.
Rathbun, S.L. 1998. Spatial modelling in irregularly shaped regions: kriging estuaries. Environmetrics 9: 109-129.
Sanders, M. 1995. Distribution of polycyclic aromatic hydrocarbons in oyster (Crassostrea virginica) and surface sediment from two estuaries in South Carolina. Archives of Environmental Contamination and Toxicology 28:397-405.
Schantz,
M.M., J.J. Nichols, and S.A. Wise. 1997. Evaluation of pressurized fluid
extraction for the extraction of environmental matrix reference materials.
Analytical
Chemistry 69: 4210-14219.
Skjemstad, J.O., L.J. Janik, M.J. Head, and S.G. Mcclure. 1993. High-energy ultraviolet photooxidation — a novel technique for studying physically protected organic-matter in clay-sized and silt-sized aggregates. Journal of Soil Science 44: 485-499.
Smayda, T.J. 1957. Phytoplankton studies in lower Narragansett Bay. Limnology and Oceanography 2: 343-359.
Swartz, R.C., D.W. Schults, and T.H. Dewitt. 1990. Toxicity of fluoranthene in sediment to marine amphipods: a test of the equilibrium partitioning approach to sediment quality criteria. Environmental Toxicology and Chemistry 9: 1071-1080.
Urbanization and Southeastern Estuarine Systems. 1994. Year 4 progress report and Year 5 research proposal. Submitted to Office of Oceanic Research Programs National Oceanic and Atmospheric Administration. March 16, 1994.
Vernberg, F.J., W.B. Vernberg, E. Blood, A. Fortner, M. Fulton, H. Mckellar, W. Michener, G. Scott, T. Siewicki, and K. El Figi. 1992. Impact of urbanization on high-salinity estuaries in the southeastern United States. Netherlands Journal of Sea Research 30: 239-248.
Vernberg, F.J. and W.B. Vernberg. 2001. The coastal zone: past, present, and future. The University of South Carolina Press, Columbia, USA.
Wirth, E.F., M.H. Fulton, G.T. Chandler, P.B. Key, and G.I. Scott. 1998. Toxicity of sediment-associated PAHs to the estuarine crustaceans, Paleomonetes pugio and A. tenuieremis. Environmental Contamination and Toxicology 61: 637-644.
Wortham, H., E.B. Nguyen, P. Masclet, and G. Mouvier. 1993. Study of heterogenous reactions of polycyclic aromatic hydrocarbons I: weakening of PAH-support bonds under photonic irradiation. The Science of the Total Environment 128: 1-11.
Address of Correspondent:
D.E. Porter
Department of Environmental Health Sciences
Arnold School of Public Health
University of South Carolina
Room 311, 900 Sumter Street
Columbia, South Carolina, USA 29208
Phone: 803.777.1465
Fax: 803.777.8769
porter@sc.edu