|
|
|
Copyright © 2001 by The Resilience Alliance
The following is the established format for referencing this article:
Kevan, P. G. and T. P. Phillips. 2001. The economic impacts of pollinator declines: an approach to assessing the consequences. Conservation Ecology 5(1): 8. [online] URL: http://www.consecol.org/vol5/iss1/art8/
A version of this article in which text, figures, tables, and appendices are separate files may be found by following this link.
Synthesis, part of Special Feature on Pollinator Decline The Economic Impacts of Pollinator Declines: An Approach to Assessing the Consequences Peter G. Kevan and Truman P. Phillips
University of Guelph
- Abstract
- Introduction
- Do Pollination Deficits Exist in Agroecosystems?
- The Economics of Pollinators
- A Model for Examining How Pollinator Deficits Affect Commodity Prices
- What Sort of Data Need to Be Collected?
- Conclusions
- Responses to this Article
- Acknowledgments
- Literature Cited
Since agricultural activities were first recorded, there have been shortages of pollinators. Today it seems that pollination systems in many areas of agriculture are threatened by the inadequacy or lack of sustainable managed, indigenous, or imported pollinators. Pollinator shortages can adversely affect crop production and commodity markets. This paper presents an economic model than can be used to measure some of the economic impacts of pollinator deficits on traded commodities. This economic analysis indicates that consumers of a commodity affected by a pollinator deficit may suffer because the commodity costs more and becomes less available. At the same time, although the producers of the affected commodity may experience crop declines, they may also experience economic gains in the form of higher prices. The amount the producer gains or loses depends on the shape of the supply and demand functions, and the magnitude of these losses or gains is an empirical question. Although there are few data available to evaluate this model, those we do have indicate that serious problems for world food supply, security, and trade could be in the offing if current declines in pollinator abundance, diversity, and availability are not reversed. Various crops and cropping systems are suggested as practical starting places for economic studies of the effects of pollinator declines, with emphasis on the type of data required.
KEY WORDS: agricultural sustainability, economics, food prices, pollinator abundance, pollinator availability, pollinator deficit, pollinator diversity, pollinator force, world food supply, trade.
Published: April 5, 2001
It is widely believed that pollination is in such serious jeopardy from the viewpoints of agricultural productivity and food security (Kevan et al. 2001) that the Convention on Biological Diversity and the Food and Agricultural Organization of the United Nations have recently (1998–2000) taken on leading roles internationally in this area. Nevertheless, little information is available on how pollination deficits affect the costs of food production. In this paper, we address the evidence for pollination deficits and discuss their extent in agroecosystems. We also present a model for assessing the costs of pollination deficits and suggest the focus for some much needed economic studies that could help to clarify this issue.
The oldest recorded examples of pollination and pollination deficit in crops are for sycomore (also known as sycamore) figs, Ficus sycomorus (Amos ca. 760 B.C.) and for date palm, Phoenix dactylifera, and Smyrna figs, Ficus carica (Herodotus 485-425 B.C.).
Although the sycomore fig is native to central eastern Africa and Yemen, it is also widely cultivated in Egypt and Mediterranean countries, where its pollinators (Ceratosolen arabicus: Hymenoptera: Agaonidae) are absent (Galil and Eisikowitch 1968, 1969a, b, 1974). Theophrastus (372-287 B.C.) recorded the lack of seeds in Egyptian figs, and Galil (1967) noted that there were no wasps associated with figs from ancient tombs. How the plant spread beyond the reaches of its pollinators is unknown, but its range included Egypt by at least 3000 B.C. (Galil 1967). For the unfertilized fruit to develop, it must be scraped in the manner described by Theophrastus (372-287 B.C.) and Galil (1967), often with a special knife (Henslow 1892, 1902, Keimer 1928). Depending on the translation, Amos (760 B.C.) describes himself variously as a fig scraper, piercer, dresser, or gatherer. Nevertheless, whatever his occupational designation, he clearly understood how to produce sycomore figs without pollinators.
The date palm is dioecious and appears to be pollinated by wind and bees (Free 1993, Roubik 1995). Because male palms are not fruitful in the sense of agricultural production, only female palms have been retained. The result, even about 3000 years ago in Mesopotamia, was that hand pollination using male inflorescences taken to the female trees was necessary (Tylor 1891, Meeuse 1981). Herodotus (485-425 B.C.) also described this practice; however, he was under the impression that it also involved a gall fly, and he mixed the techniques used for the anthropogenic pollination of dates and F. carica. Pollination was probably associated with festivals of spring and fertility in the region at the time of the Prophet Mohammed, who reportedly discouraged such festivals and only reluctantly recognized the need to hand-pollinate dates (Margoliouth 1905: 230, Fraser 1935: II: 25, V: 281). The best pollination results today are obtained by tying dehiscent staminate inflorescences into the pistillate inflorescences of female palms (McGregor 1976, Mbaya and Kevan 1995), or by other artificial means.
The need for caprification in Smyrna figs, i.e., providing production trees with a pollinating wasp containing caprifigs, was known in ancient Greece (Herodotus 485-425 B.C.) and Turkey (Condit 1920, Goor 1965). In addition, both Herodotus (485-425 B.C.: I: 193) and Aristotle (350 B.C.: 26) had some understanding of the role of wasps in pollination, although they referred to the insects involved as gall flies and psene, respectively. By the mid-18th century, the process of pollination was better understood in figs according to Knuth (1909: III: 372), who reported that even Linnaeus spoke of a special "messenger of love" needed to fertilize Smyrna figs. However, this concept was vociferously ridiculed in Europe (Reasoner 1891) and California (Condit and Swingle 1947) in the mid- and late 1800s, so it is not hard to understand why the Smyrna figs introduced into California in the late 1800s failed to bear fruit. It was not until Eisen (1891) introduced the wasps into California that fruit set was achieved. However, problems persisted because of the lack of caprifig fruit; these were eventually overcome when the U.S. Department of Agriculture (USDA) implemented a program to provide such figs to growers (Rixford 1918). McGregor (1976) provides a synopsis of this story.
At about the same time as the fig pollination problem was being resolved in California, the shortage of pollinators for seed production of red clover, Trifolium pratense, in New Zealand prompted the introduction of bumble bees, Bombus spp., from Europe (Belt 1876, Dunning 1886, Hopkins 1914). Their establishment was successful, although New Zealand has still not solved its on-going problems with regard to the pollination of kiwifruit, Actinidia deliciosa (Free 1993, Roubik 1995).
More recently, Malaysia, where labor costs for hand pollination are rising sharply, found a solution to its shortage of pollinators for oil palm, Elaeis guineensis. Syed (1979) studied the pollination of this important crop plant in its native West Africa and worked out the relationship between the pollinating weevils, Elaeidobius spp., and the inflorescences of the male and female palms. After careful screening and quarantine, Elaeidobius kamerunicus was released in Malaysian oil palm plantations, where it rapidly became established and spread (Syed et al. 1982). The result continues to be the sustainable and sufficient pollination of crops whose harvests exceed those previously produced by hand pollination, with savings of millions of U.S. dollars per year (Kevan et al. 1986).
Another example of placing pollinators into a novel habitat to enhance crop production is the introduction of bumble bees into hothouses to pollinate tomatos, Lycopersicon esculentum, in Europe (Banda and Paxton 1991) and North America (Kevan et al. 1991). Artificial pollination with electric vibrators (Kerr and Kribs 1955) is a costly method that is no longer used, whereas buzz pollination (Buchmann 1983) by bumble bees produces superior fruit (Banda and Paxton 1991, Kevan et al. 1991). Morandin (2000) describes the efforts being made to solve the remaining technological problems related to hothouse pollination. The value of "bombiculture" for producing hothouse tomatos and other fruit has not been assessed, but must amount to millions of dollars worldwide.
Although it may be argued that these examples are special cases and that the pollinator deficits were artificial, they serve to illustrate that, when pollinator forces are insufficient, there may be inexpensive, effective alternative methods of solving problems related to pollinator deficits.
The success story of the alfalfa leaf-cutter bee (Megachile rotundata) and its culture for the pollination of alfalfa (Medicago sativa), both exotic organisms in North America, is well known. The pioneering work of Bohart (1972) and Hobbs (1967) has given rise to the multimillion-dollar industry of "megachileculture," whose huge economic benefits are described by Olmstead and Woolen (1987). Bohart (1957) also recognized the problem of providing adequate pollination to alfalfa seed production fields, which led to the commercial development of practices for encouraging and maintaining pollinators other than honey bees, especially the alkali bee (Nomia melanderia). On the Canadian prairie, problems with alfalfa pollination and concomitant seed yield declines can be attributed to the expanding agriculture of the 1940s, when fields were larger and more kempt. As a result of the subsequent reduction in nesting habitat, there were too few native pollinators to provide pollination for any plants except those at the peripheries of large fields (Peck and Bolton 1946, Stephens 1955). In Manitoba, Stephens (1955) recorded yields of 1000 kg/ha from small fields, but only 15 kg/ha from large fields. In Ontario, the contemporaneous decline of alfalfa seed production has been attributed to changing agricultural practices, including the use of insecticides.
Habitat destruction has also been a problem in the pollination of cacao (Theobroma cacao). Overly fastidious management of plantations included the removal of rotting vegetation, the substrate in which the pollinating midges undergo larval development (Winder 1977), and yield reductions ensued. By purposely placing appropriate plant material such as banana (Young 1982) or palm trunks (Ismail and Ibrahim 1986), adequate pollinator forces can be encouraged and maintained. The destruction of Brazilian habitat for pollinators of Brazil nuts (Bertholletia excelsa) has been even more detrimental to production (Maués 2001). Brazil nuts are pollinated by an assemblage of large bees whose nesting habitats have been severely curtailed or even eliminated (Sutton and Collins 1991). The vicious cycle of habitat destruction or pollution, paucity of pollinators, and failures in plant reproduction, recruitment, and regeneration has been well described (Janzen 1974, Kevan 1974, 1975a).
The catastrophic effects of recently introduced parasitic mites on honey bees have changed the face of apiculture in North America. Colony mortality and intensive management have made it more expensive to keep bees. The number of beekeepers has declined, as has the number of colonies being kept all over North America. Other pests also threaten to make beekeeping more costly and difficult. Pollination has been adversely affected, and growers have reported difficulties in obtaining services for crops such as blueberries in Maine, pome fruit in the northeastern United States and Canada, almonds in California, field cucumbers in the eastern United States and Canada, and hybrid seed production in western Canada. Economic analyses of the effects of parasitic mites are much needed for beekeeping per se and for the ancillary benefits of pollination (Morse and Calderone 2000). Although the epidemic of Nosema that reportedly swept through the cultures of Bombus occidentalis used in hothouse tomato pollination on the Pacific coast of North America has had major repercussions, the economic consequences have not been analyzed.
The adverse effects of pesticides on pollinators are well understood, especially from a toxicological viewpoint (Johansen and Mayer 1990), although less is known about their impact on crop reductions. Several works (Kevan 1975b, 1977, Kevan and LaBerge 1979, Kevan and Oppermann 1980, Kevan and Plowright 1995, Kevan et al. 1999) explore the effects of applications of the organophosphorous pesticide Fenitrothion on nontarget habitat and on blueberry pollinators in New Brunswick, Canada. The demise of the pollinators resulted in such severe declines in the blueberry crop in the affected regions that provincial yields were significantly below those of neighboring Nova Scotia and Maine (Kevan 1977, Kevan and Oppermann 1980, Kevan and Plowright 1995), with an annual harvest loss of about 0.75 x 106 kg. Intensity of agricultural activities has also been shown to correlate with lower (by about 50%) populations and diversity of pollinators in apple orchards in British Columbia (Scott-Dupree and Winston 1987) and berry production areas (MacKenzie and Winston 1984). Kevan (1999) presents more details about these and other examples, but crop yields have rarely been included in such studies.
Although the economic impacts of pollinator declines have not been well recorded, we think it can be safely assumed that many local economies are being affected. Examples of available studies of this type include Siebert (1980), who provides an estimate of the revenue losses to both almond growers and honey producers in California resulting from a pesticide-induced decline in the numbers of pollinators; Olmstead et al. (1987), who document the historical and economic effects of the addition of pollinators on the production of alfalfa seed; and Cox et al. (1991), who show that the demise of fruit bats (Megachiroptera) through overhunting in South Pacific islands has reduced the pollination and fruit yields of some traditional harvests. No matter what their cause, would we expect anything different to result from pollinator declines elsewhere?
Although several works have attempted to illustrate the severity of pollinator declines (Buchmann and Nathan 1996, Matheson et al. 1996, Kearns et al. 1998, Kevan 1999), the problem has generally been ignored. For this reason, it is appropriate to ask the following questions from the point of view of documentation: "Are pollinator declines real?" and "Do they have economic consequences for agriculture?" We would not only answer both questions in the affirmative, but we also believe that the problem is extremely serious, with far-reaching consequences for agriculture and global food production. However, even the most obvious example of honey bee pests and diseases should be carefully examined. The previous example of the demise of pollinators on lowbush blueberry heaths in New Brunswick shows how the basic pollination biology of crops is linked, through production agriculture and ecosystem health, to economic impacts on consumers (Belaoussoff and Kevan 1998). Issues of scale are important. For example, even though the demise of pollinators on lowbush blueberry production fields in New Brunswick adversely affected yields, farm-gate income, and other aspects of the local economy (casual employment), it did not affect the overall market price for blueberries because that was set elsewhere by broader, regional effects (Kevan and Oppermann 1980). Below we explore the broader context of inter-regional or international trade.
The inadequacy of pollinator forces for agricultural production can be offset by providing services through imported pollinators, encouraging local populations to grow, or both. However, cost-benefit analyses for pollination services in agriculture are not readily available. Some cropping systems that would lend themselves well to such studies are alfalfa seed production, hothouse tomato production, some small berry and tender fruit crops, field cucumbers and melons, almonds and other orchard crops, and specialty production systems (e.g., hybrid and horticultural seed production).
The value of pollination to alfalfa seed growers in the Canadian prairies is estimated to be 35% of annual crop production (Blawat and Fingler 1994), although the usual practice there is for seed growers and custom pollination providers or "megachileculturalists" to share the risks, costs, and benefits in various ways. In Saskatchewan, Manitoba, and Alberta, about 30,000 ha of pedigree alfalfa seed was grown each year in 1999 and 2000, with yields of about 200–800 ha/kg worth Can.$0.50–0.75/kg. Based on a calculation of area (30,000 ha) x yield (300 kg/ha) x value (Can.$0.60 kg) x 35%, the value per year of pollination services to the production of pedigree alfalfa seed in these provinces amounted to about Can.$2 million. Pedigree alfalfa seed is grown on about one-third of the total acreage under alfalfa seed production, but common seed has a lower market value. It is reasonable to place the value of pollinators to the alfalfa seed industry at about Can.$6 million per year in the Canadian prairies, whereas the value of megachileculture (including bees for export, specialized equipment, etc.) in Saskatchewan alone, which has about half its prairie acreage in alfalfa seed production, is estimated at Can.$10 million (W. Goertzen, personal communication). Olmstead and Woolen (1987) estimated that, when pollination services were provided to Utah over the period they were studying, the increase in alfalfa production amounted to about a 600% return on investment. These values are representative of the scale of the value of pollination, although a detailed economic analysis based on the different farming systems (e.g., dryland vs. irrigation) has yet to be carried out.
When studying apple production in Ontario, Kevan (1997) calculated roughly that providing about one hive of honey bees per hectare resulted in about one extra seed per apple, which produced larger and more symmetrical apples. These improved apples were estimated to provide marginal returns of about 5–6%, or about Can.$250/ha, compared to an orchard without honey bees. The cost of pollination services at that time was about 1% of production costs, and the greater yield represented a return to the grower of 700% of the cost of pollination services. Cane (1996) assessed the value of individual wild bees (Habropoda laboriosa) as pollinators of rabbiteye blueberry (Vaccinium ashei) at about U.S.$20.00. These models represent valuable and practical approaches to evaluating pollinators as an agricultural production cost with huge potential benefits. Unfortunately, the economics of bombiculture and hothouse tomato production seem to have been set artificially by the high cost of the alternative of hand pollination.
Pollinator deficits have multiple effects on the price of a commodity. In economic terms, the price of that commodity reflects the costs of production, distribution, and marketing plus profit. Thus pollinator deficits may increase the cost of production as the cost of providing pollinator services rises, owing to the greater demand for that service. Pollinator deficits may also cause a shift in the supply function, which may in turn result in a higher market price.
The economic impacts of pollinator deficits occur at various levels in the consumption-production continuum. At one extreme, there are the individuals who bear the brunt of the impact of pollinator deficits. For example, individual producers may experience a complete crop failure and resulting economic losses, or individual consumers may not be able to consume a particular commodity because pollinator deficits have made it unavailable. At the other extreme, there is the aggregate market, which may show little change in the total amount supplied by producers or demanded by consumers. In practice, a pollinator decline can be expected to affect both individuals and the market.
At the market level, basic economic theory considers a pollinator deficit that causes a reduction in production to be a shift in the supply function. This shifted supply function and the existing demand function lead to a new prevailing price. Normally the new price is higher than the original price, and the equilibrium quantity is less.
Southwick and Southwick (1992) provide a more detailed approach to the economics of pollination (specifically of honey bees to U.S. agriculture), covering the issues of supply and demand, costs, and the effects of changes in yields. Their model allows for the assessment of the effects of pollinator declines on the overall market. It is understood that, if commodity producers as a group (throughout areas where the commodity is produced) adopt the same practices for better yields, supply will increase and prices will drop.
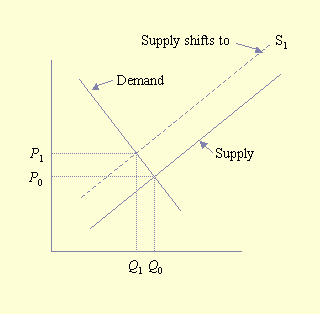
Their model for the economic analysis of pollinator deficits is not conceptually difficult. The simplest economic model is one in which production and consumption occur in a closed system, i.e., one in which products are not traded. In this simple economic model, equilibrium exists when supply equals demand, at which time the price of goods is represented as P0 and the quantity consumed is Q0 (Fig. 1). Conceptually, the effect of a pollinator deficit is to shift the supply curve (S) to the left, producing a new supply curve, S1. This shift results in a new equilibrium position to the northwest of the original equilibrium. At this new position, less product is consumed, Q1, at a higher price, P1. The unequivocal impact of a pollinator deficit is that consumers are worse off because they consume less at a higher price. The impact on producers is less clear: less product is sold, but it is sold at a higher price. In the case of an inelastic demand function (i.e., a vertical demand curve), producers would actually benefit financially from a pollinator deficit because the amount sold would not change, whereas the price received would increase. Conversely, for an elastic demand curve (i.e., a horizontal demand curve), producers would be severely harmed by a pollinator deficit. The net impact on producers and consumers depends on the elasticity (slope and shape) of the supply and demand functions.
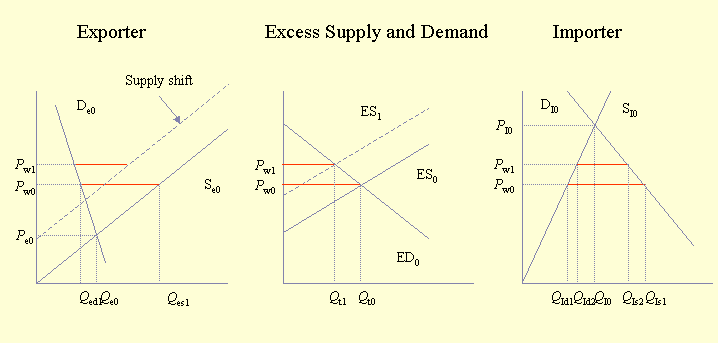
The economic impact of pollinator deficits on traded agricultural commodities has received little attention. Furthermore, the global aspects of pollination management, forces, and deficits have not been examined. The economic impact of pollinator deficits described above can be expanded to include the effects on traded commodities. With its three panels, Fig. 2 illustrates what happens when two countries or regions trade with each other. The left panel represents the country with the lower domestic price for a particular commodity, whereas the right panel represents the country with the higher domestic price for that commodity. Trade occurs between lower and higher priced sources of the commodity. The amount traded and the traded price can be determined from the central panel of excess supply and demand. The excess supply curve represents the difference between the exporter's supply and demand curves. The excess demand curve represents the difference between the importer's demand and supply curves. We also provide a more detailed presentation of Figures 2-5 to illustrate the interactions between prices and quantities and between supply and demand when trade is considered. Readers using Internet Explorer should view this version. Readers using Netscape should view this version.
|
Fig. 3. Measurement of consumer and producer surplus: an example. P represents price, and Q represents quantity.

|
|
Fig. 4. Consumer and producer surpluses for an importing country (pollinator deficit in the exporting country). Please refer to Figs. 2 and 3 for definitions of terms.
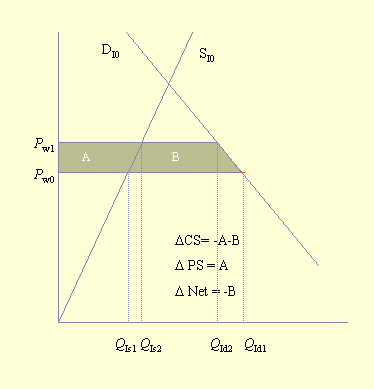
|
|
Fig. 5. Consumer and producer surpluses for an exporting country with a pollinator deficit. Please refer to Figs. 2 and 3 for definitions of terms.
|
The initial situation is that trade occurs between the two countries at price Pw0, and the amount traded is Qt0. The impact of trade on the exporting country with the lower price (left-hand panel) is to raise the price from Pe0 to Pw0, decrease the amount consumed domestically from Qe0 to Qed1, and increase the amount produced from Qe0 to Qes1. The impact of trade in the country with higher prices (right-hand panel) is the opposite: the price drops, the amount consumed locally increases, and the amount produced locally decreases.
From this starting point we can examine the impact of pollinator deficits. A deficit in the country with lower prices shifts the supply curve to the left, which in effect narrows the gap between national production and consumption. In the middle panel, this is equivalent to moving the excess supply curve to the left. The impact of this movement is to decrease trade and to increase the traded price of the commodity. The impact of a pollinator deficit is that consumers in both countries lose, and producers in the country with higher prices gain because they are able to supply more at a higher price. Producers in the country with lower prices may lose or gain; as depicted in Fig. 2, they appear to lose. Depending on the actual supply and demand functions in the country with lower prices, it is possible that producers gain because the proportional increase in price is larger than the proportional decline in production.
The alternative scenario is that of a pollinator deficit in the (importing) country with higher prices. In this case, the supply curve in this country would shift to the left, causing the excess demand curve to shift to the right and up. As a result, total trade would increase, and the traded price would also increase. The obvious winner would be the producers in the country with lower prices. Other potential winners are traders, who would benefit from increased trade, and producers in the country with lower prices would gain if the price increase were greater than the decrease in the amount produced.
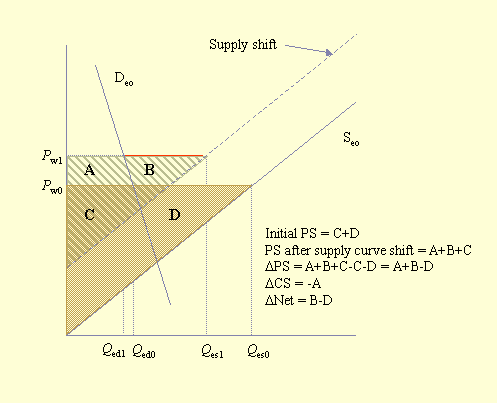
Southwick and Southwick (1992) demonstrated how to measure these benefits and losses. However, although the general economic approach is to measure what economists call "consumer surpluses" and "producer surpluses," Southwick and Southwick considered only consumer surpluses. Figure 3 illustrates consumer and producer surpluses in an equilibrium situation. The consumer surplus is the area under the demand curve and above the equilibrium price, triangle CS. This area, a measure of consumer welfare, is considered to be a surplus and is defined as the "... excess of the price that the consumer would be willing to pay for each unit consumed over the price which is actually paid ..." (Sadoulet and de Janvry 1995: 189). The producer surplus is the area above the supply curve and below the equilibrium price, triangle PS. It represents the excess returns that the producer receives for each unit of production below the price that is actually paid (Sadoulet and de Janvry 1995). The economic impact of an event such as a pollinator deficit can be measured by the change in the consumer and producer surpluses. The societal impact of this event is measured as the sum of the consumer and producer surpluses.
Figure 4 illustrates the impact of a pollinator deficit on the importing country. As shown in Fig. 2, the price of the commodity increases from Pw0 to Pw1, and the amount consumed deceases from QId1 to QId2. Finally, domestic production increases from QIs1 to QIs2. Thus, producers experience an increase (A) in the producer surplus, and the area above the supply curve increases by the same amount. On the other hand, consumers experience a loss of -A-B, and the area below the demand curve decreases by that amount. The net societal change is a welfare loss of -B. This confirms the earlier observation that producers in the importing country benefit and consumers lose when a pollinator deficit is experienced in an exporting country.
The impact of a pollinator deficit in the exporting country, while conceptually the same as for the importing country, is more complicated to assess. Figure 5 illustrates the impact on consumer and producer surpluses for the exporting country (the one with lower prices) shown in Fig. 2. The shift of the supply curve to the left, owing to pollinator decline, results in consumers paying more for less product and producers selling less at a higher price. The producer surplus changes from area C + D to area A + B + C. Thus, the net change is (A + B + C) - (C + D) = A + B - D. The loss in consumer surplus is the area A, and the net societal change is the area B - D. Depending on the relative sizes of B and D, the net societal gain could be positive or negative.
These illustrations of the economic impact of pollinator declines show that, in all cases, the consumer is hurt, whereas producers who have not experienced a pollinator deficit may benefit. These results apply only to a situation in which unrestricted trade in a commodity can occur between two or more countries. Barriers to trade will affect the magnitude of the economic impact of a pollinator deficit. Furthermore, the magnitude of the losses or gains is an empirical problem that has two components: the shape of the supply and demand functions and data availability. A discussion of the estimation of supply and demand curves is outside the scope of this paper, but the estimation of these curves is dependent on the collection of appropriate data. Clearly, a key element in understanding the impacts, both physical and economic, of pollinator declines is the collection of appropriate data.
There is a body of information on the value of pollination to agriculture, including a review of the value of honey bees as pollinators of U.S. crops (Morse and Calderone 2000). Other data of that sort have been accumulated for Canada, Australia, New Zealand, the United Kingdom, and elsewhere. Pollination has even been worked into attempts to evaluate the value of nature's services to humankind (Costanza et al. 1997, Nabhan and Buchmann 1997). However, all those estimates are based on considerable guesswork.
Although a small body of information on the value of pollination to particular crops, especially alfalfa seed, is available, there is an even greater volume of information on the effects of pollination on crop quality. The latter has not been, but could be, converted into monetary units (Free 1993, Roubik 1995, Cane 1996, Kevan 1997, Delaplane and Mayer 2000). The concept of the economic pollinator force also requires more consideration (Kevan et al. 1986). If pollinators are free, then an excess of pollination is of no concern except under special circumstances. If pollinators are costly, then the grower needs to know that the marginal benefits of expanding the pollinator force are at least as great as the marginal costs. Individual farmers and grower groups at local and regional levels could make good use of information about the potential for greater harvests through better pollination. Fortunately, some databases containing information on the costs and uses of pollinator services are being developed in the Pacific Northwest and California (Burgett 1995). Nevertheless, there is a need for more studies that provide economic analyses of the relationships among pollinators, relevant crop production, and the market.
What types of data are needed to assess the potential, and possibly real, production and economic effects of pollinator deficits? We suggest the following, with comments on the general availability of each type.
Although there is some information on pollination requirements (Free 1993, Roubik 1995, Delaplane and Mayer 2000), much of it should be completely re-evaluated because it is dated, anecdotal, and not based on scientific methods. With regard to the most useful pollinators and their effects on crop yields, all too often honey bees are advocated as the only practical pollinators. However, it is well known that, for some crops, including several mentioned above, this is simply not the case (Parker et al. 1987, Kevan et al. 1990, Torchio 1990, Westerkamp 1991, Batra 1995, Cane 1996, Kevan 1999). In addition, assemblages of pollinators provide the most powerful pollination forces for other crops (Kevan et al. 1997, Melendez 1997); obviously more research is needed in both these areas. With regard to cost effectiveness, the value of improved crops must be set against the cost of better pollination, and the cost of pollination in terms of the overall cost of producing particular crops is likely to vary widely.
Although these problems at first seem daunting, we could learn a lot from crops whose pollination requirements and value to growers are already well understood, such as apples, blueberries, cranberries, field cucumbers and melons, squash and pumpkin, hothouse tomatos, almonds, oil palm, and cacao.
To fully analyze the economic impact of pollinator deficits, it would be necessary to include alternative commodities that may be influenced by the change in market conditions (Southwick and Southwick 1992). However, it is clear that commodity information on production, costs of production, prices, sales, and trade, as well as information on the incidence and severity of pollinator deficits represent core data for the economic analysis of the impact of pollinator deficits.
Of course, the degree of uncertainty of these conclusions would increase with the expanding scale of consideration.
We conclude that there is ample information to suggest the existence of pollinator declines that have affected, and are affecting, agricultural productivity. We have also shown that, when trade occurs, these declines have different economic impacts on producers and consumers in importing and exporting countries. The analysis attempts to illustrate that the full economic impact of pollinator declines cannot be determined by examining only the decrease in production that is caused by pollinator declines.
The magnitude of pollinator declines can be determined by empirical analysis. We note that few examples allow for much analysis, but there are some in which crop quantity and quality could be converted into monetary units (e.g., blueberries, palm oil, cranberries, pome fruits, squash and pumpkin, field cucumbers) with some additional research. Clearly there is a need for pollination ecologists to join forces with economists who share their interests in production agriculture and provide more comprehensive analyses of this problem. Such research could yield information that would enable individual farmers to integrate pollination into their fiscal and land-use planning. While the pollination requirements of many crops appear to be small, their marginal impact may be large. The adverse economic effects of pollinator deficits on food prices must follow from on-farm considerations, but have extensive ramifications for world food supply, security, and trade.
Responses to this article are invited. If accepted for publication, your response will be hyperlinked to the article. To submit a comment, follow this link. To read comments already accepted, follow this link.
We thank the organizers of the Workshop on "Causes and Extent of Declines among Native Invertebrate Pollinators: Detection, Evidence and Consequences" held at the National Center for Ecological Analysis and Synthesis on October 10–12, 1999, in Santa Barbara, California. We thank the staff at NCEAS for their help and hospitality. We are grateful to W. Goertzen (Saskatchewan Alfalfa Seed Producers' Association), G. Hubner (Manitoba Forage Seed Producers' Association), and L. Lyzenga (Alberta Alfalfa Seed Producers Association) for insights into their industry. We also thank our co-participants in the workshop for their generous sharing of ideas. Carlos Greco kindly read our manuscript and suggested improvements. Finally, we thank two reviewers who provided valuable suggestions.
Amos. ca. 760 B.C. Amos 7: 14 in The Holy Bible containing the Old and New Testaments translated out of the original tongues and with former translations diligently compared and revised by His Majesty's Special Command [King James' version]. Eyre and Spottiswoode, London, UK.
Aristotle. ca. 350 B.C. Historia animalium. Republished in 1910 in D'Arcy Wentworth Thompson, translator. The works of Aristotle translated into English. Volume IV. Oxford University Press, London, UK.
Banda, H. J., and R. J. Paxton. 1991. Pollination of greenhouse tomatoes by bees. Acta Horticulturae 288: 194-198.
Batra, S. W. T. 1995. Bees and pollination in our changing environment. Apidologie 26: 361-370.
Belaoussoff, S., and P. G. Kevan. 1998. Toward an ecological approach for the assessment of ecosystem health. Ecosystem Health 4: 4-8.
Belt, T. 1876. Bees and clover. Nature (London) 13: 26.
Blawat, P., and B. Fingler. 1994. Guidelines for estimating cost of production: alfalfa seed. Farm Business Management Information Update. Manitoba Agriculture, Winnipeg, Manitoba, Canada.
Bohart, G. E. 1957. Pollination of alfalfa and red clover. Annual Review of Entomology 2: 355-380.
Bohart, G. E. 1972. Management of wild bees for the pollination of crops. Annual Review of Entomology 17: 287-312.
Buchmann, S. E. 1983. Buzz pollination in angiosperms. Pages 73-113 in C. E. Jones and R. J. Little, editors. Handbook of experimental pollination biology. Van Nostrand Reinhold, New York, New York, USA.
Buchmann, S. E., and G. P. Nabhan. 1996. The forgotten pollinators. Island Press, Washington, D.C., USA.
Burgett, M. 1995. Pacific Northwest Honey Bee Pollination Survey. American Bee Journal 136: 432-434.
Cane, J. H. 1996. Lifetime monetary value of individual pollinators: the bee Habropoda laboriosa at rabbiteye blueberry (Vaccinium ashei Reade). Proceedings of the Sixth International Symposium on Vaccinium Culture, Orono, Maine, USA, August 12-17, 1996. Acta Horticulturae 446: 67-70.
Condit, I. J. 1920. Caprifigs and caprification. California Agriculture Experiment Station Bulletin 319: 341-375.
Condit, I. J., and W. T. Swingle. 1947. The fig. Chronica Botanica, Waltham, Massachusetts, USA.
Costanza, R., R. D'Arge, R. de Groot, S. Farber, M. Grasso, B. Hannon, K. Limburg, S. Naeem, R. V. O'Neill, J. Paruelo, R. G. Rifkin, O. Sutton, and M. van den Belt. 1997. The value of the world's ecosystem and natural capital. Nature (London) 387: 253-260.
Cox, P. A., T. Elmquist, E. Pierson, and W. E. Rainey. 1991. Flying foxes as strong interactors in South Pacific island ecosystems: a conservation hypothesis. Conservation Biology 5: 448-454.
Delaplane, K. S., and D. F. Mayer. 2000. Crop pollination by bees. CAB International, Wallingford, UK.
Dunning, J. W. 1886. The importation of humble bees into New Zealand. Transactions of the Royal Entomological Society, London 6: 32-34.
Eisen, G. 1891. The first introduction of Blastophaga psenes into California. Insect Life 4: 128-129.
Fraser, J. G. 1935. The golden bough. Third edition. MacMillan, New York, New York, USA.
Free, J. B. 1993. Insect pollination of crops. Second edition. Academic Press, London, UK.
Galil, J. 1967. Sycomore wasps from ancient Egyptian tombs. Israel Journal of Entomology 2: 1-9.
Galil, J. 1968. An ancient technique for ripening sycomore fruit in east-mediterranean countries. Economic Botany 22: 178-190.
Galil, J., and D. Eisikowitch. 1968. On the pollination ecology of Ficus sycomorus in East Africa. Ecology 49: 259-269.
Galil, J., and D. Eisikowitch. 1969a. Further studies on the pollination ecology of Ficus sycomorus L. (Hymenoptera, Chalcidoidea, Agaonidae). Tijdschrift voor Entomologie 112: 1-13.
Galil, J., and D. Eisikowitch. 1969b. Note on pollen transport, pollination and protection of ovaries in Ficus sycomorus. New Phytologist 68: 1243-1244.
Galil, J., and D. Eisikowitch. 1974. Further studies on the pollination ecology of Ficus sycomorus. II. Pocket filling and emptying by Ceratosolen arabicus Mayr. New Phytologist 73: 515-528.
Goor, A. 1965. The history of the fig in the Holy Land from ancient times to the present. Economic Botany 19: 124-135.
Henslow, G. 1892. Egyptian figs. Nature (London) 47: 102.
Henslow, G. 1902. The sycomore fig. Journal of the Royal Horticultural Society London 27: 128-131.
Herodotus. 485-425 B.C. Historiae. Republished in G. Rawlinson, translator. Wordsworth classics of world literature. Ware, UK.
Hobbs, G. A. 1967. Domestication of alfalfa leaf-cutter bees. Canada Department of Agriculture Publication Number 1313, Ottawa, Ontario, Canada.
Hopkins, I. 1914. History of the bumblebee in New Zealand—its introduction and results. New Zealand Department of Agriculture, Industry and Commerce Bulletin Number 46 (new series), Christchurch, New Zealand.
Ismail, A., and A. G. Ibrahim. 1986. The potential for ceratopogonid midges as insect pollinators of cocoa in Malaysia. Pages 471-484 in M. Y. Hussein and A. G. Ibrahim, editors. Biological control in the tropics. Universiti Pertanian Malaysia, Serdang, Selangor, Malaysia.
Janzen, D. H. 1974. The de-flowering of Central America. Natural History 83: 48-53.
Johansen, C. A., and D. F. Mayer. 1990. Pollinator protection: a bee and pesticide handbook. Wicwas Press, Cheshire, Connecticut, USA.
Kearns, C. A., D. W. Inouye, and N. M. Waser. 1998. Endangered mutualisms: the conservation of plant-pollinator interactions. Annual Review of Ecology and Systematics 29: 83-112.
Keimer, L. 1928. An ancient Egyptian knife in modern Egypt. Ancient Egypt 1928: 65-66.
Kerr, E. A., and W. Kribs. 1955. Electric vibrator as an aid in greenhouse tomato production. Queensland Journal of Agricultural Science 2: 157-169.
Kevan, P. G. 1974. Pollination, pesticides, and environmental quality. BioScience 24: 198-199.
Kevan, P. G. 1975a. Pollination and environmental conservation. Environmental Conservation 2: 293-298.
Kevan, P. G. 1975b. Forest application of the insecticide Fenitrothion and its effects on wild bee pollinators (Hymenoptera: Apoidea) of lowbush blueberries (Vaccinium spp.) in southern New Brunswick, Canada. Biological Conservation 7: 301-309.
Kevan, P. G. 1977. Blueberry crops in Nova Scotia and New Brunswick—pesticides and crop reductions. Canadian Journal of Agricultural Economics 25: 61-64.
Kevan, P. G. 1997. Honeybees for better apples and much higher yields: study shows pollination services pay dividends. Canadian Fruitgrower (May 1997): 14, 16.
Kevan, P. G. 1999. Pollinators as bioindicators of the state of the environment: species, activity and diversity. Agriculture, Ecosystems and Environment 74: 373-393.
Kevan, P. G., E. A. Clark, and V. G. Thomas. 1990. Insect pollinators and sustainable agriculture. American Journal of Alternative Agriculture 5: 13-22.
Kevan, P. G., C. F. Greco, and S. Belaoussoff. 1997. Log-normality of biodiversity and abundance in diagnosis and measuring of ecosystemic health: pesticide stress on pollinators on blueberry heaths. Journal of Applied Ecology 34: 1122-1136.
Kevan, P. G., N. Y. Hussein, N. Hussey, and M. B. Wahid. 1986. Modelling the use of Elaeidobius kamerunicus for pollination of oil palm. Planter (Malaysia) 62: 89-99.
Kevan, P. G., V. Imperatriz-Fonseca, G. W. Frankie, C. O'Toole, C. H. Vergara, and T. Feltz, editors . 2001. Pollinating bees: the conservation link between agriculture and nature. International Bee Research Association, Cardiff, UK, in press.
Kevan, P. G., and W. E. LaBerge. 1979. Demise and recovery of native pollinator populations through pesticide use and some economic implications. Pages 489-508 in Proceedings of the Fourth International Symposium on Pollination, Maryland Agricultural Experiment Station Special Miscellaneous Publication Number 1, College Park, Maryland, USA.
Kevan, P. G., and E. B. Oppermann. 1980. Blueberry production in New Brunswick, Nova Scotia and Maine: reply to Wood et al. Canadian Journal of Agricultural Economics 28: 81-84.
Kevan, P. G., and R. C. Plowright. 1995. Impact of pesticides on forest pollination. Pages 607-618 in J. A. Armstrong and W. G. H. Ives, editors. Forest insect pests in Canada. Natural Resources Canada, Canadian Forest Service, Science and Sustainable Development Directorate, Ottawa, Ontario, Canada.
Kevan, P. G., W. A. Straver, M. Offer, and T. M. Laverty. 1991. Pollination of greenhouse tomatoes by bumblebees in Ontario. Proceedings of the Entomological Society of Ontario 122: 15-19.
Knuth, P. 1909. Handbook of flower pollination: based upon Hermann Müller's work 'The fertilization of flowers by insects.' Volume III. Observations on flower pollination made in Europe and the arctic regions on species belonging to the natural orders Goodenovieae to Cycadeae. Oxford University Press, London, UK.
MacKenzie, K. E., and M. L. Winston. 1984. Diversity and abundance of native bee pollinators of berry crops and natural vegetation in the Lower Fraser Valley, British Columbia. Canadian Entomologist 116: 965-974.
Margoliouth, D. S. 1905. Mohammed and the rise of Islam. Third edition. Putnam, New York, New York, USA.
Matheson, A., S. L. Buchmann, C. O'Toole, P. Westrich, and I. H. Williams, editors. 1996. The conservation of bees. Linnean Society Symposium Series Number 18. Academic Press, London, UK.
Maués, M. M. 2001. Reproductive phenology and pollination of the Brazil Nut Tree (Bertholletia excelsa Humb. & Bonpl. Lecythidaceae) in eastern Amazonia. In P. G. Kevan, V. Imperatriz-Fonseca, G. W. Frankie, C. O'Toole, C. H. Vergara, and T. Feltz, editors. Pollinating bees: the conservation link between agriculture and nature. International Bee Research Association, Cardiff, UK, in press.
Mbaya, J. K. S., and P. G. Kevan. 1995. Applied pollination in Africa. Pages 57-62 in D. W. Roubik, editor. Pollination of cultivated plants in the tropics. FAO, Food and Agriculture Service Bulletin Number 118, Rome, Italy.
McGregor, S. E. 1976. Insect pollination of cultivated crop plants. U.S. Department of Agriculture Handbook Number 496, Washington, D.C., USA.
Meeuse, B. J. D. 1981. The story of pollination. Ronald Press, New York, New York, USA.
Meléndez Ramírez, V. 1997. Polinización y biodiversidad de abejas nativas asociadas a cultivos hortícolas en el estado de Yucatán, México. Thesis. Universidad Autónoma de Yucatán, Facultad de Medicina Veterinaria y Zootécnica, Mérida, México.
Morandin, L. A. 2000. Bumble bee (Bombus impatiens) pollination of greenhouse tomatoes. Thesis. University of Western Ontario, London, Ontario, Canada.
Morse, R. A., and N. W. Calderone. 2000. The value of honey bees as pollinators of U.S. crops in 2000. Bee Culture (March 2000): 2-15.
Nabhan, G. P., and S. E. Buchmann. 1997. Services provided by pollinators. Pages 133-150 in G. C. Daily, editor. Nature's services: societal dependence on natural ecosystems. Island Press, Washington, D.C., USA.
Olmstead, A., and D. B. Woolen. 1987. Bee pollination and productivity growth: the case of alfalfa. American Journal of Agricultural Economics 69: 56-63.
Parker, F. D., S. W. T. Batra, and V. Tepedino. 1987. New pollinators for our crops. Agricultural Zoology Review 2: 279-304.
Peck, O., and J. L. Bolton. 1946. Alfalfa seed production in northern Saskatchewan as affected by bees, with a report on the means of increasing the populations of native bees. Scientific Agriculture 26: 338-418.
Reasoner, P. W. 1891. The condition of tropical and semi-tropical fruits. U.S. Department of Agriculture, Division of Pomology, Bulletin Number 1, Washington, D.C., USA.
Rixford, G. P. 1918. Smyrna fig culture. U.S. Department of Agriculture Bulletin Number 732, Washington, D.C., USA.
Roubik, D. W., editor. 1995. Pollination of cultivated plants in the tropics. FAO, Food and Agriculture Service Bulletin Number 118, Rome, Italy.
Sadoulet, E., and A. de Janvry. 1995. Quantitative development policy analysis. Johns Hopkins University Press, Baltimore, Maryland, USA.
Scott-Dupree, C. D., and M. L. Winston. 1987. Wild bee diversity and abundance in orchard and uncultivated habitats in the Okanagan Valley, British Columbia. Canadian Entomologist 119: 735-745.
Siebert, J. W. 1980. Beekeeping, pollination and externalities in California agriculture. American Journal of Agricultural Economics 62: 165-171.
Southwick, E. E., and L. Southwick Jr. 1992. Estimating the economic value of honey bees (Hymenoptera: Apidae) as agricultural pollinators in the United States. Journal of Economic Entomology 85: 621-633.
Stephen, W. P. 1955. Alfalfa pollination in Manitoba. Journal of Economic Entomology 48: 543-548.
Sutton S. L., and N. M. Collins. 1991. Insects and tropical forest conservation. Pages 405-422 in N. M. Collins and J. A. Thomas, editors. The conservation of insects and their habitats. Academic Press, London, UK.
Syed, R. A. 1979. Studies on oil palm pollination. Bulletin of Entomological Research 69: 213-224.
Syed, R. A., I. H. Law, and R. H. V. Corley. 1982. Insect pollination of oil palm: introduction, establishment and pollinating efficiency of Elaeidobius kamerunicus in Malaysia. Planter (Malaysia) 58: 547-561.
Theophrastus. 372-287 B.C. Peri phyton historia. Republished in 1961 in A. Hart, translator. Inquiring into plants. Heinemann, London, UK.
Torchio, P. F. 1990. Diversification of pollination strategies for U.S. crops. Environmental Entomology 19: 1694-1656.
Tylor, E. B. 1889. Fertilization of date palm in ancient Assyria. Academy (London) 35: 396.
Westerkamp, C. W. 1991. Honeybees are poor pollinators—why? Plant Systematics and Evolution 177: 71-75.
Winder, J. A. 1977. Some organic substrates which serve as insect breeding sites in Bahian cocoa plantations. Review of Brazilian Biology 37: 351-356.
Young, A. M. 1982. Effect of shade cover and availability of midge breeding sites on pollinating midge populations and fruit set in two cacao farms. Journal of Applied Ecology 19: 149-155.
Address of Correspondent:
Peter G. Kevan
Department of Environmental Biology
University of Guelph
Guelph, Ontario, Canada N1G 2W1
Phone: (519) 824-4120 ext.2479
Fax: (519) 837-0442
pkevan@uoguelph.ca