|
|
|
Copyright ©1999 by The Resilience Alliance*
Ås, S. 1999. Invasion of matrix species in small habitat patches. Conservation Ecology [online] 3(1): 1. Available from the Internet. URL: http://www.consecol.org/vol3/iss1/art1/
A version of this article in which text, figures, tables, and appendices are separate files may be found by following this link.
Research Invasion of Matrix Species in Small Habitat Patches Stefan Ås
Department of Animal Ecology, Evolutionary Biology Center, Uppsala University, Sweden
- Abstract
- Introduction
- Material and Methods
- Results
- Discussion
- Responses to This Article
- Acknowledgments
- Literature Cited
The diversity, expressed as species richness in equal-sized samples, of wood-living beetles in successional stages of deciduous forest after forest fires approximately 100 years ago did not differ between patch sizes in this study. Thus, the crucial question for conservation is whether the lack of reduction in diversity in small patches means that a number of small patches can elevate the regional diversity as efficiently as can one larger area. In the present study, the smaller patches did not differ from larger patches in substrate availability, quality, or heterogeneity. The frequency of a group of species was measured as the number of occurrences, viz. the number of trees on which a species was found, summed over all species in the group. The number of occurrences of species unique to the specific patch-size group did not differ between large and small patches. The number of occurrences of species that preferred matrix areas (managed coniferous forest and clearcuts) was, however, larger than expected in small patches. The results indicate that small patches, although having high local (alpha) diversity, are more similar in species composition to neighboring matrix areas (reduced beta diversity). Thus, small areas contribute less to the regional (gamma) diversity than do larger areas.
KEY WORDS: alpha (local) diversity, beta (between-habitats) diversity, deciduous forest, disturbance, diversity, fire, gamma (regional) diversity, invasion, matrix habitat, patch size, species-richness.
Published February 10, 1999.
Diversity is a key element in conservation, both for its own sake and as an approximation of an area's biological importance when precise knowledge of the species composition, or the present threat status of the involved species, is lacking (Wilson and Willis 1975). In many cases, its use as an approximation is probably a justifiable strategy, but carries a risk of losing specialized species restricted to species-poor areas.
Diversity is a complex term that sometimes is used synonymously with species richness. However, diversity also includes evenness of the distribution of individuals on species. As with most ecological terms, species diversity has scale-dependent properties. A useful classification of these properties is the subdivision of diversity into three categories (alpha, beta, and gamma diversity), as proposed by Whittaker (1972). In this scheme, alpha diversity is the local diversity within a habitat, beta diversity is a function of the difference between habitats, and gamma diversity is the diversity over a geographical range including a number of different habitat patches, viz. regional diversity. Cody (1975), however, defines gamma diversity as a function of the change in species composition in similar habitats in different geographical areas. To avoid confusion, I use the term regional diversity for gamma diversity, sensu Whittaker (1972). If the goal of conservation is to preserve maximal species diversity within a region (regional diversity), then this goal can be attained by either maximizing the diversity within each patch or increasing the difference in patch properties and, hence, the variation of species composition among patches (Murphy 1989). An important complement to information on the alpha species diversity of a patch is therefore what contribution this patch gives to the regional species diversity (Andrén 1994).
It is not surprising that species richness and diversity on an island or in a habitat patch, in most cases, will increase with island or patch area. This relationship has been studied for many decades (Ås et al. 1992, 1997) and has been seen as one of the few genuine laws of modern community ecology (Schoener 1976). However, many critical studies have found no difference in diversity between different-sized areas, and indeed, examples of the reverse relationship also exist ( e.g., MacGarvin 1982, Lathi and Ranta 1985, Loman and von Schantz 1991, Robinson et al. 1992, Vickery et al. 1994).
Although the diversity in a patch depends on its area, a less isolated patch may have increased diversity by invasion from surrounding habitats, compared to a more isolated patch of similar size. I use the term invasion because I include both normal immigration and mass effect, i.e., the establishment of species in sites where they are not self-maintaining (Shmida and Wilson 1985). The size of the different patches also might influence invasions, and a smaller patch should be more susceptible to invasions than a large one (Saunders et al. 1991, Hobbs and Huenneke 1992, Halme and Niemelä 1993). The impact of species from surrounding habitats is of great importance, especially on habitat islands (e.g., Janzen 1983, 1986, Shmida and Wilson 1985, Wilcox and Murphy 1985, Andrén and Angelstam 1988, Angelstam 1992, Ås 1993, Andrén 1994). Smaller patches may have greater alpha diversity than larger patches of the same habitat because of invasions. The increased similarity between the small patches and surrounding habitats (reduced beta diversity) may lead simultaneously to reduced regional diversity if only small patches are preserved.
If smaller patches are depauperated in terms of species richness, we should expect a difference in diversity between equal-sized samples from large and small patches. I have shown in a previous study that this is not necessarily the case for beetles living in dead wood. The diversity of wood-living beetles in small patches (<20 ha) of deciduous forest does not differ from the diversity in larger areas (>100 ha) of similar habitat (Ås 1993). The diversity in this investigation was measured as the residual from the regression of the log-transformed number of species as a function of the logarithm of stem area examined of all dead trees rooted within the sampling plots. This measure of diversity is area independent, and to distinguish between diversity per unit area and total diversity, I will refer to the former as relative diversity. It seems unlikely that the lack of significant reduction in relative diversity is merely a consequence of inadequate sampling, because the mean value from small areas actually exceeds that from larger areas. These patches of deciduous forest are successions after forest fires that occurred approximately 100 years ago, and are distributed in an island-like manner in a conifer-dominated boreal forest landscape. In order to evaluate the relative contribution of large and small patches to regional diversity, I have reanalyzed these data to explore possible reasons for the lack of reduction of relative diversity in small patches.
The hypothesis I test is: invasion and establishment of species from the surrounding habitats explain the lack of reduction in relative diversity in small areas. I also test two alternative hypotheses proposed to explain this pattern: (1) habitat differences between small and large areas explain the observed pattern, and (2) smaller areas harbor more specialized species. If true, the second hypothesis may be attributed to either specialization for small areas per se, or, more likely, specialization for edge habitats. If many species are specialized for edge habitats, then small patches would have higher than expected diversity as a consequence of having a larger proportion of their area as edges. Larger patches will, of course, have a larger total edge area, which will probably harbor a larger total number of species. In relative terms, however, small areas might contain more species than expected if the edge is much more species-rich per unit area than the core.
All areas examined in this investigation are situated in the middle boreal zone (Ahti et al. 1968) of central Sweden, were burned in the late 1880s, and have since been unaltered by man. Samples were also taken in the surrounding managed forest and from small patches of mostly deciduous forest in the vicinity of the areas. The dominating deciduous tree species in all areas are two birch species, Betula pendula and B. pubescens. Approximately 65-70% of the broadleaf trees belong to these species. Aspen (Populus tremula) is the third most abundant species, with 25-30% of all individuals. Goat willow (Salix caprea), gray alder (Alnus incana), and rowan (Sorbus aucuparia) occur regularly but sparsely. The surrounding coniferous forest is dominated by Scots pine (Pinus sylvestris) on drier soils, whereas Norway spruce (Picea abies) dominates in more productive sites. Modern forestry has converted practically all forested areas into managed coniferous forest that are homogenous, both in age structure and in species composition. The surrounding coniferous forests, which later will be referred to as the matrix, thus consist of both clearcuts and mature coniferous forest, and all age classes in between. All of these areas are, to different degrees, exposed to different silvicultural actions such as thinning or removal of deciduous trees. A more detailed description of the study areas can be found in Ås (1993).
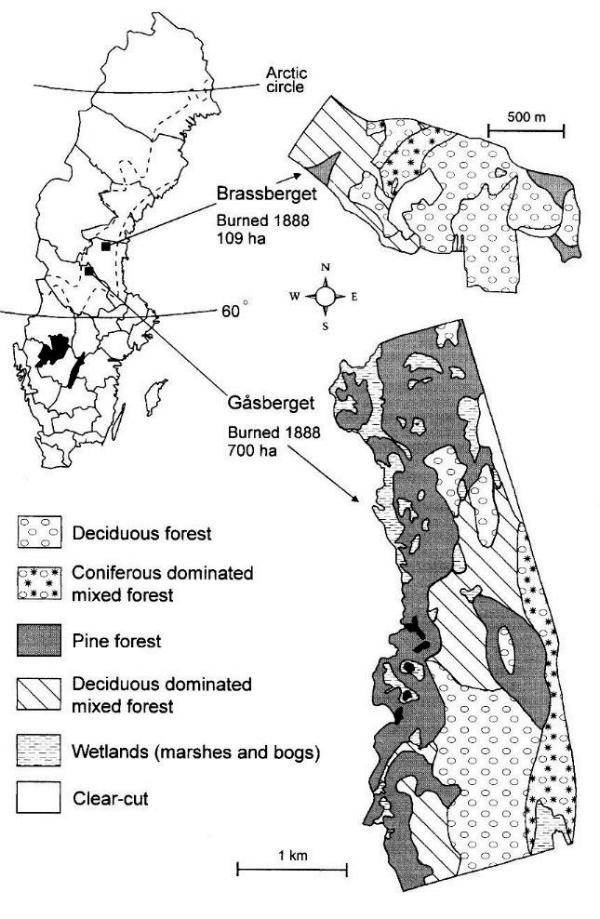
Gåsberget (60o18' N, 15o20' E) is the largest of the areas and is situated in the northeast corner of the county of Dalarna, Sweden. Intense forestry has converted most of the natural forests to managed coniferous forests in this landscape. The deciduous forest at Gåsberget originated in 1888 after a fire that burned an area of approximately 4000 ha (Lundquist 1986). Of this area, 700 ha of naturally revegetated forest has been undisturbed, whereas the rest has been converted to managed coniferous forest, except for a few scattered, small patches of mainly deciduous mixed stands (Fig. 1). In the northern part of the originally burned area, 4 km north of Gåsberget, there is a smaller (<5 ha) deciduous-dominated forest, called Päjertjärn, that resembles the deciduous-dominated mixed-forest patches within the Gåsberget area, in terms of species composition in the tree and field layers (Fig. 1).
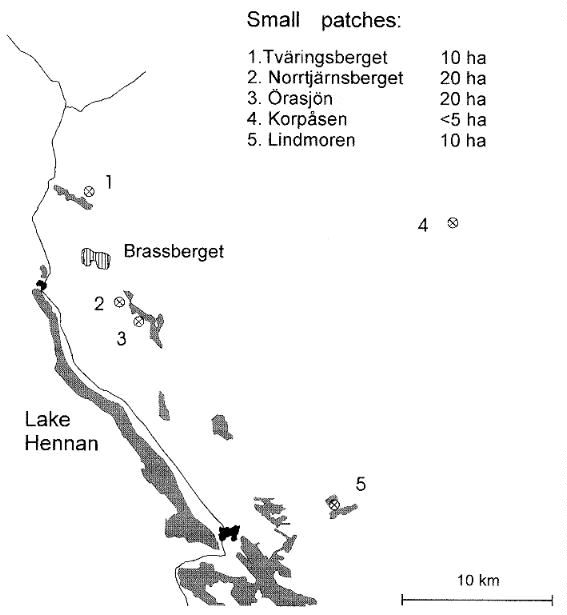
The other study area, Brassberget (62o12' N, 15o47' E), is in the northwestern part of the county of Hälsingland. The forests surrounding this area have not been as intensively managed as those around Gåsberget, and the proportion of deciduous trees and areas with old forest is accordingly higher. The high proportion of deciduous trees and the commonness of fire scars show that the whole region has had frequent fires in past centuries. The forest in and around Brassberget had burned as often as, on average, once every 34 yr before the last substantial fire that occurred in 1888 (Sturesson 1983). This fire covered an area of approximately 460 ha, of which a core area of 109 ha has been left for natural regeneration. This area is now dominated by deciduous forest and is surrounded by coniferous forest and clearcuts (Fig. 1). Five smaller areas, ranging from 5 to 20 ha, of deciduous forest in the vicinity of Brassberget were also examined. Four of them originated after fires, and one is a reforestation of previously cultivated land (Korpåsen, Fig. 2). The species composition of the trees and of the field layer in these areas are very similar to those in the deciduous forest in Brassberget, although Lindmoren is somewhat lusher, with scattered individuals of more southerly species such as linden Tilia cordata.
Substrate availability for beetles living in dead deciduous trees in small and large areas of deciduous forest, measured as the mean basal area of dead deciduous trees (standing and lying) per hectare, did not differ. Large areas of deciduous forest had a basal area of dead deciduous trees of 6.8 ± 0.62 m2/ha (mean ± 1 SE), whereas the corresponding figure for the smaller patches was 7.0 ± 0.98 m2/ha (for a more detailed description and statistical evaluation, see Ås 1993).
Sampling methodTo determine the species composition within large (>100 ha) and small (<20 ha) areas of deciduous-dominated forest, I investigated a number of sampling plots on three or four occasions. On each sampling date, a 50 x 5 m rectangle within the deciduous dominated forest was examined. From every dead tree (standing or lying) that was, or had been, rooted within the rectangle, I removed bark, cambium, and all loose material between 1 and 1.5 m from the root. All this material, including bark and cambium, was then sieved, and the animals were extracted in Tullgren funnels within 24 hours. Because of the very low density of dead trees in the matrix areas, i.e., managed coniferous forest, a different sampling technique was used. Because a 50 x 5 m area would contain very few dead trees, approximately 20 dead deciduous trees first encountered along a transect were chosen, but each tree was examined in the same way as in the plots. Each 50 x 5 m rectangle, or transect, could be sampled only once, due to the sample technique used. Therefore, on the next occasion, samples were taken from areas next to the one previously sampled. The analyses in this paper are only based on data from the most abundant deciduous tree species, birch (Betula pendula and B. pubescens), aspen, goat willow, and gray alder.
Ten plots were sampled within the Gåsberget area, six in the largest deciduous forest patch, and one in each of the four smaller patches of deciduous forest. One plot was sampled in the Päjertjärn area. All plots at Gåsberget and Päjertjärn were sampled on three different occasions, May and August 1987 and May 1988. In the surrounding managed forest, five sampling plots were selected at random, two in cleared areas and three in mature coniferous forests, all within the originally burned 4000-ha area. Matrix samples were taken in May and August 1988.
In the Brassberget area, three of the five sampling plots were situated in pure deciduous forest. The fourth was located in a pure deciduous stand within the pine-deciduous mixed forest, and the fifth plot was in the spruce-deciduous forest. In the smaller patches of deciduous forest near Brassberget, the two larger ones (Örasjön and Norrtjärnsberget) were sampled with two plots, whereas the three smaller ones (Lindmoren, Korpåsen, and Tväringsberget) contained one each. As at Gåsberget, five plots of managed forest near Brassberget were sampled. Three were in mature coniferous forest and two in clear-cut areas. All plots, except those in managed forest, were sampled in May and August 1987 as well as in 1988. The plots in managed forest were only sampled in May and August 1988.
The material from 1987 and 1988 was pooled in the analyses, because the ranked abundances of the different species were significantly correlated between years. This is true for the two large areas as well as for the combined small areas (Ås 1993).
On each sampling occasion, I measured the amount and quality of resources available for wood-living beetles. The resource amount was measured as the basal area of dead deciduous trees per hectare, whereas the estimates of substrate quality consisted of measurements of the state of decay and the abundance of tree fungi. Decay was estimated for each trunk and divided into four classes: (1) fresh wood; (2) bark starting to fall off, some soft wood; (3) large parts of the trunk with soft wood; and (4) almost no hard wood at all. The amount of fungal infection was classified in a similar fashion: (1) no sign of fungi; (2) sparse occurrence of mycelium under bark or in wood; (3) rich occurrence of mycelium under bark or in wood, with some (1-3) fruiting bodies; and (4) rich occurrence of mycelium and many (>3) fruiting bodies. Because each plot was sampled on three or four different dates and because each new sample was taken in a new 50 x 5 m rectangle close to the previously sampled one, estimations of both means and spatial variability could be made.
The nomenclature of the beetles follows Lundberg (1986), and a more detailed description of the sampling method can be found in Ås (1993).
There is a risk in using the number of individuals of different species when examining the relative abundance of wood-living beetle species because of their clumped distribution. For this reason, the number of occurrences, calculated as the number of trees on which a species was found within a plot, was used as a quantitative estimate of the relative densities of the different species. Few species were found to occur more often in either small or large areas (four and five species, respectively), and only 56 of the 155 species occurred in sufficiently high numbers to allow an analysis of their distribution (Ås 1993). Therefore, the sum of occurrences for species found only in large or only in small areas, respectively, weighted by the total number of occurrences, was used to analyze whether small patches had more occurrences of specialized species than did larger areas.
Ås (1993) found that 13 species occurred more often than expected by chance in managed forest (including clearings). These species were: Tachys nanus (Caribidae), Scaphisoma agaricinum (Scaphidiidae), Gabrius exspectatus, Atrecus pilicornis, Acrulia inflata, Dinaraea aequata, and D. linearis (Staphylinidae), Bibloporus minutus (Pselaphidae), Denticollis linearis and Ampedus nigrinus (Elateridae), Corticaria rubripes (Latridiidae), Cis boleti (Cisidae), and Schizotus pectinicornis (Pyrochroidae). The number of occurrences summed over these species, in large and small patches, respectively, was used to test if the lack of reduction in relative diversity is caused by invasions from matrix habitats.
In total, 3997 beetles of 155 species were collected. Of these, 1630 individuals belonging to 104 species were found in large areas of deciduous forest, and 773 individuals from 83 species were found in smaller deciduous forest patches. The matrix areas sampled in 1988 yielded 1594 individuals of 119 species (a complete species list is available from the author on request or at http://www.zoologi.uu.se/zooeko/stefan/home.html).
To test whether smaller patches offered a more favorable habitat for wood-living beetles, I compared mean substrate availability and quality in small patches to that of larger areas of deciduous forest. I also predicted that a more variable patch would be inhabited by more species. Because it is not obvious whether a higher degree of decay, or fungal infection, should be regarded as high or low quality, the test for these variables is two-tailed. All other tests are one-tailed with H0 : small areas have equal or lower values than large areas. However, there were no differences between small and large areas in any of the variables examined (Wilcoxon two-sample tests; see Table 1).
|
Table 1. Difference between sample plots in large areas of
deciduous forest (Size = L, n = 15) and smaller patches of
deciduous forest (Size = S, n = 8) in the amount and quality of
substrate available for wood-living beetles. Because each plot was
sampled with three or four 5 x 50 m rectangles, the mean value of each
variable is used. The standard deviation is used as a measurement of
the spatial variability of the variable. The differences between the
two size groups were examined with Wilcoxon two-sample tests. *Two- tailed tests; all other tests are one-tailed, with H0 = S <= L.
|
Consistent differences in tree species composition between large and small habitat patches could also influence the result. I compared the mean stem area examined of the different tree species per plot in large and small patches, using MANOVA and a t test for the separate tree species. Neither the MANOVA (Wilks' lambda = 0.832, P = 0.33) nor the t test (Table 2) revealed any significant difference between plots from the different-sized habitat patches.
Table 2. Mean stem area examined per plot of the dominant deciduous tree species from large and small patches of deciduous forest. Numbers within parentheses are standard errors. The differences between patch-size groups were examined with t tests. No correction for unequal variance was used because no significant difference in variance was found.
|
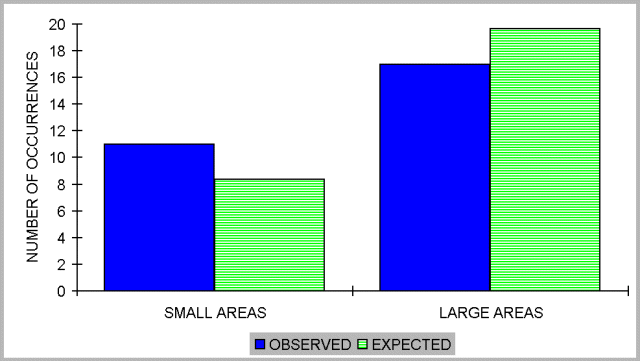
The sum of the number of occurrences over all species was 647 (Ol) in samples from larger areas, whereas it was 275 for small patches (Os). Species unique to larger areas accounted for 17 of these occurrences (Ul), and 11 occurrences were due to species unique to smaller patches (Us). Hence, the expected number of unique occurrences for large areas was (Ul+Us)Ol/(Ol+Os)=19.65, and for small patches was (Ul+Us)Os/(Ol+Os)=8.35. Large and small areas did not differ in the number of occurrences of unique species (chi-square = 1.234, P = 0.27: Fig. 3).
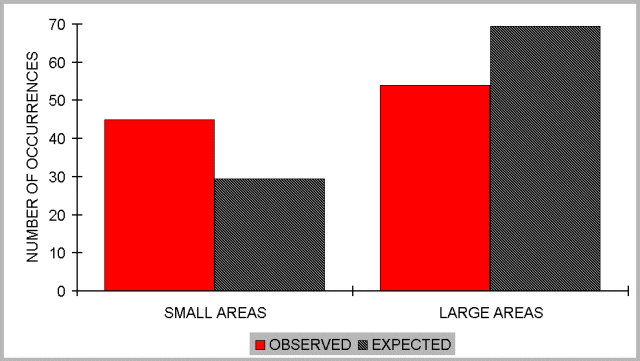
If the reason for the enhanced diversity within smaller patches (i.e., the lack of reduced diversity as expected from a species-area relation) was invasion of species from the surrounding managed forest, we should expect a relatively higher proportion of occurrences of these species in the smaller patches compared to the larger areas. This prediction was supported. The number of occurrences summed over species preferring matrix habitats was 45 (Ms) in smaller patches and 54 (Ml) for larger areas. The expected number of occurrences for smaller patches was (Ml + Ms)Os/(Ol + Os)=29.53 and for larger areas (Ml + Ms)Ol/(Ol + Os)=69.47. There were significantly more occurrences in samples from smaller patches than from larger areas (chi-square = 12.947, P = 0.001; Fig. 4).
Preservation of the largest possible number of native species in a region is an obvious goal of conservation (Murphy 1989). This goal of maximizing regional diversity can only be achieved by maintaining alpha or beta diversity, or both. It is, however, possible that there is a trade-off between alpha and beta diversity. For example, consider a mosaic of habitats that is replaced with the most diverse habitat type. When converting patches of different habitats to resemble the most species-rich habitat type, most of the species that will be added to the local fauna are species that were already present in the existing patches of the most species-rich habitat. On the other hand, species that were specific to the habitat under conversion may vanish. The end result of this conversion is that each patch will gain species, but the region as a whole will become less diverse (Andrén 1994, Davis 1994). In other words, alpha diversity will increase in each patch, but beta diversity simultaneously will decrease and the net result will be a reduced regional diversity.
The relative increase in diversity in small patches can, at least temporarily, be even more exaggerated if the small-sized patches are results of a fragmentation process. When the size of an area is restricted because of modification of the surrounding area, it may initially have a large fraction of the original species present. Due to low population sizes, many of these species may go extinct, causing a relax to a number, the size of which depends on area (MacArthur and Wilson 1967). At the same time, the area is exposed to invasion of species from the altered surrounding areas. If the relaxation in the number of species that was originally present is slow relative to the invasion of new species, then the diversity may increase initially as a consequence of the fragmentation (Verner 1986, Murphy 1989, Webb 1989, Harris and Scheck 1991, Saunders et al. 1991). This phenomenon may occur in my study area, because most of the intensive forestry actions responsible for the fragmentation of the deciduous forest are fairly recent (< 50 yr; Esseen et al. 1992, 1997, Ås 1993).
The invasion of species into a small patch is an edge effect. The species that will gain from a fragmentation of larger habitat patches are those that thrive in edge habitats, independent of whether or not the edge is their prime habitat (Andrén 1992, Angelstam 1992, Hobbs and Huenneke 1992). It is true that a larger patch will have a larger edge area, which probably will harbor more species than the smaller edge area from a smaller patch. In a landscape with a given amount of habitat, however, fragmentation into small patches will lead to an increase of the total amount of edge area. The relative proportion of edge-thriving species will therefore increase, probably with negative impact on the species restricted to the patches. If small patches are more species-rich than expected, and this increase in species number has been caused by species that are specialized on edge habitats, then small patches could increase the regional diversity, depending on whether edge habitat was common or rare before fragmentation (Andrén 1994). In the present case, however, a more probable cause of a higher than expected relative diversity of wood-living beetles in small patches is the addition of species from the managed matrix forest which, hence, will reduce the beta diversity.
If invasion from the matrix is the prime reason for the presence of extra species in the edge, an edge from a large patch is probably not equal to an edge from a small patch, in terms of species composition. Assume that population density and mean dispersal distances are equal for patch and matrix species. If patch size were considerably larger than the mean dispersal distance of an individual, a dead tree at the edge of a patch would get approximately 50% of all new individuals from within the patch. With decreasing patch area, a diminishing part of the area within the dispersal range would be patch habitat. As a consequence, a larger amount of all individuals reaching the dead tree will originate from the matrix habitat.
In the case of wood-living beetles in boreal-zone deciduous trees, a great part of the high diversity in the small patches of deciduous forest is clearly due to invasions by species from the managed forest. Most of these species mainly inhabit clear-cut areas. This is not surprising, because disturbance by forest fires and windstorms, under pristine conditions, frequently creates large, open patches with abundant substrate available for a limited time. This type of habitat should favor species with good colonization ability. Likewise, these species should also be superior in invading edge habitats in the vicinity.
In this experiment, different-sized patches did not differ in habitat quality or in the occurrence of unique species. It seems reasonable to assume that smaller patches become more similar to matrix areas in their species composition over time and, thus, contribute less to the regional species diversity than do larger areas. Because of the relatively short time since the fragmentation of these deciduous forests, it is possible that the loss of specialized species from small patches might be exacerbated with time. On the other hand, these small patches are probably important in a landscape context. They may act as sink areas that increase the population size of species otherwise restricted to larger areas (Pulliam 1988, Danielson 1992, Dunning et al. 1992). They may also increase the landscape connectivity and, as a result, facilitate colonization of new, large patches of deciduous-dominated forest (Taylor et al. 1993). These positive effects of small areas thus depend on the presence of larger areas, which need to be preserved to maintain regional diversity.
Responses to this article are invited. If accepted for publication, your response will be hyperlinked to the article. To submit a comment, follow this link. To read comments already accepted, follow this link.
I thank J. Bengtsson, T. Ebenhard, S. Ulfstrand, and Stanly H. Faeth for valuable comments on the manuscript, and L.-O. Wikars for assisting in the field and identifying the numerous specimens, together with R. Baranowski. This work has been financially supported by the Swedish Environmental Protection Agency and the Crafoord Foundation.
Ahti, T., L. Hämet-Ahti, and J. Jalas. 1968. Vegetation zones and their sections in northwestern Europe. Annales Botanici Fennici 5:169-211.
Andrén, H. 1992. Corvid density and nest predation in relation to forest fragmentation: a landscape perspective. Ecology 73:794-804.
_______ . 1994. Effects of habitat fragmentation on birds and mammals in landscapes with different proportions of suitable habitat: a review. Oikos 71:355-366.
Andrén, H., and P. Angelstam. 1988. Elevated predation rates as an edge effect in habitat islands. Ecology 69:544-547.
Angelstam, P. 1992. Conservation of communities: the importance of edges, surroundings and landscape mosaic structure. Pages 9-70 in L. Hansson, editor. Ecological principles of nature conservation. Elsevier, London, UK.
Ås, S. 1993. Are habitat islands islands? Wood-living beetles (Coleoptera) in deciduous forest fragments in boreal forest. Ecography 16:219-228.
Ås, S., J. Bengtsson, and T. Ebenhard. 1992. Archipelagoes and theories of insularity. Pages 201-251 in L. Hansson, editor. Ecological principles of nature conservation. Elsevier, London, UK.
Ås, S., J. Bengtsson, and T. Ebenhard. 1997. Archipelagoes and theories of insularity. Ecological Bulletins 46:88-116.
Danielson, B. J. 1992. Habitat selection, interspecific interactions and landscape composition. Evolutionary Ecology 6:399-411.
Davis, A. L. V. 1994. Habitat fragmentation in southern Africa and distributional response patterns in five specialist or generalist dung beetle families (Coleoptera). African Journal of Ecology 32:192-207.
Dunning, J. B., B. J. Danielson, and H. R. Pulliam. 1992. Ecological processes that affect populations in complex landscapes. Oikos 65:169-175.
Esseen, P.-A., O. Ehnström, L. Ericson, and K. Sjöberg. 1992. Boreal forests - the focal habitats of Fennoscandia. Pages 252-325 in L. Hansson, editor. Ecological principles of nature conservation. Elsevier, London. UK.
Esseen, P.-A., O. Ehnström, L. Ericson, and K. Sjöberg. 1997. Boreal forests. Ecological Bulletins 46:16-47.
Halme, E. and J. Niemelä. 1993. Carabid beetles in fragments of coniferous forest. Annales Zoologici Fennici 30:17-30.
Harris, L. D., and J. Scheck. 1991. From implications to applications: the dispersal corridor principle applied to conservation of biological diversity. Pages 189-220 in D. A. Saunders and R. J. Hobbs, editors. Nature conservation 2: the role of corridors. Surrey Beatty and Sons, Chipping Norton, Australia.
Hobbs, R. J., and L. F. Huenneke. 1992. Disturbance, diversity, and invasion: implications for conservation. Conservation Biology 6:324-337.
Janzen, D. H. 1983. No park is an island: increase in interference from outside as park size decreases. Oikos 41:402-410.
_______ . 1986. The eternal external threat. Pages 286-303 in M. E. Soulé, editor. Conservation biology. The science of scarcity and diversity. Sinauer Associates, Sunderland, Massachusetts, USA.
Lathi, T., and E. Ranta. 1985. The SLOSS principle and conservation practice: an example. Oikos 44:369-370.
Loman, J., and T. von Schantz. 1991. Birds in a farmland - more species in small than in large habitat islands. Conservation Biology 5:176-188.
Lundberg, S. 1986. Catalogus Coleopterorum Suecicae. Entomologiska Föreningen, Naturhistoriska Riksmuseet, Stockholm, Sweden.
Lundquist, R. 1986. Gåsberget - en skogsbiologisk inventering i Kopparbergs län. Länsstyrelsen i Kopparbergs län, Falun, Sweden. (In Swedish.)
MacArthur, R. H., and E. O. Wilson. 1967. The theory of island biogeography. Princeton University Press, Princeton, New Jersey, USA.
MacGarvin, M. 1982. Species-area relationships of insects on host plants: herbivores on Rosebay willowherb. Journal of Animal Ecology 51:207-223.
Murphy, D. D. 1989. Conservation and confusion: wrong species, wrong scale, wrong conclusions. Conservation Biology 3:82-84.
Pulliam, H. R. 1988. Sources, sinks, and population regulation. American Naturalist 132:652-661.
Robinson, G. R., R. D. Holt, M. S. Gaines, S. P. Hamburg, M. L. Johnson, H. S. Fitch, and E. A. Martinko. 1992. Diverse and contrasting effects of habitat fragmentation. Science 257:524-526.
Saunders, D. A., R. J. Hobbs, and C. R. Margules. 1991. Biological consequences of ecosystem fragmentation: a review. Conservation Biology 5:18-32.
Schoener, T. W. 1976. The species-area relation within archipelagoes: models and evidence from land birds. Pages 629-692 in H. J. Frith and J. H. Calaby, editors. Proceedings XVI International Ornithological Congress (1974). Australian Academy of Science, Canberra, Australia.
Shmida, A., and M. V. Wilson. 1985. Biological determinants of species diversity. Journal of Biogeography 12:1-20.
Sturesson, E. 1983. Naturskogen på Brassberget - en skogsbiologisk inventering. Länsstyrelsen Gävleborgs län, Gävle, Sweden. (In Swedish.)
Taylor, P. D., L. Fahrig, K. Henein, and G. Merriam. 1993. Connectivity is a vital element of landscape structure. Oikos 68:571-573.
Verner, J. 1986. Predicting effects of habitat patchiness and fragmentation - the researcher's viewpoint. Pages 327-329 in J. Verner, M. L. Morrison, and C. J. Ralph, editors. Wildlife 2000: modeling habitat relationships of terrestrial vertebrates. University of Wisconsin Press, Madison, Wisconsin, USA.
Vickery, P. D., M. L. Hunter, Jr., and S. M. Melvin. 1994. Effects of habitat area on the distribution of grassland birds in Maine. Conservation Biology 8:1087-1097.
Webb, N. R. 1989. Studies on the invertebrate fauna of fragmented heathland in Dorset, U.K., and the implications for conservation. Biological Conservation 47:153-165.
Whittaker, R. H. 1972. Evolution and measurements of species diversity. Taxon 21:213-251.
Wilcox, B. A., and D. D. Murphy. 1985. Conservation strategy: the effects of fragmentation on extinction. American Naturalist 125:879-887.
Wilson, E. O., and E. O. Willis. 1975. Applied biogeography. Pages 522-534 in M. L. Cody and J. M. Diamond, editors. Ecology and evolution of communities. Harvard University Press, Cambridge, Massachusetts, USA.
Address of Correspondent:
Stefan Ås
Department of Animal Ecology
Evolutionary Biology Center
Uppsala University
Norbyvägen 18D
SE-752 36 Uppsala
Sweden
Phone: (46) 18 471 29 35
Fax: (46) 18 471 64 84
Stefan.As@zoologi.uu.se
*The copyright to this article passed from the Ecological Society of America to the Resilience Alliance on 1 January 2000.