|
|
|
Copyright ©1998 by The Resilience Alliance*
Elisabet Lindgren. 1998. Climate and tickborne encephalitis. Conservation Ecology [online] 2(1): 5. Available from the Internet. URL: http://www.consecol.org/vol2/iss1/art5/
A version of this article in which text, figures, tables, and appendices are separate files may be found by following this link.
Research Climate and Tickborne Encephalitis Elisabet Lindgren
Stockholm University
- Abstract
- Introduction
- Methods
- Results
- Discussion
- Speculation
- Responses
- Acknowledgments
- Literature Cited
Climatic changes are projected to alter the abundance, dynamics, and geographical distribution of many vector-borne diseases in human populations. Tick-borne diseases such as Lyme disease and tick-borne encephalitis (TBE) are a growing concern in northern Europe and the United States. The impact of a future climate change on the transmission of tick-borne diseases is not known. To make such assumptions, more empirical data are needed on the relations between short-term fluctuations in contemporary weather and disease incidence. This paper analyzes relations between daily minimum and maximum temperatures, monthly precipitation, and TBE incidence during a 36-yr period in Stockholm County, a high-endemic region for TBE in Sweden. Multiple regression analyses were performed, with temperature variables expressed as number of days per winter or spring - summer - fall season with temperatures above, below, or in the interval between different temperature limits. The limits used for daily minimum temperatures represent bioclimatic thresholds of importance for pathogen transmission. To adjust for the length of the tick's life cycle, each TBE incidence rate was related to meteorological data over two consecutive years. Results reveal that increased incidence of tick-borne encephalitis is related to a combination of two successive years of more days with temperatures permitting prolonged seasonal tick activity and, hence, pathogen transmission (i.e., daily minimum temperatures above 5ºC-10ºC), and a mild winter preceding the year before the incidence year (i.e., fewer winter days with minimum temperatures below -7ºC). Alternative explanations of the results are discussed. Findings of this study suggest that a climate change may extend the seasonal range and intensify the endemicity of tick-borne diseases, in particular, at northern latitudes.
KEY WORDS: Encephalitis; tickborne; Tickborne diseases; Temperature; Climate; Ticks.
Climatic changes are expected to have an impact on the incidence, seasonal range, and geographical distribution of several vector-borne diseases in humans, including malaria, dengue fever, lymphatic filariasis, and several arboviruses (Levins et al. 1994, Matsuoka and Kai 1994, Reeves et al. 1994, Martens et al. 1995, 1997, Martin and Lefebvre 1995, IPCC 1996a, McMichael et al. 1996, Patz et al. 1996).
The human contribution to the atmospheric concentration of greenhouse gases is predicted to cause an increase in global mean temperature of 1 - 3.5ºC by late in the next century (IPCC 1996b). The magnitude of alterations in temperature and precipitation patterns will differ regionally. The largest increases in surface temperatures are predicted to occur at high northern latitudes (Maskell et al. 1993, IPCC1996b). Over the next 50 yr, the annual mean temperature is expected to increase by 1.5 - 2.5ºC in the northern parts of Europe and the United States (Hadley Center 1995).
Ticks are the main human disease vectors at higher northern latitudes. Tick-borne diseases are a growing concern in both Europe and the United States. The different symptoms of what was later to be named Lyme disease were described in Europe during the turn of this century. A link to tick bites was suggested during the 1920s and 1930s, but the disease did not come into focus until after an outbreak in Connecticut in the nid-1970s, and the causative pathogen was identified some years later. During the 1990s, the incidence of Lyme disease increased rapidly in the Northern Hemisphere. Tick-borne encephalitis (TBE) is caused by a flavivirus with at least two subtypes: the Central European subtype and the Far Eastern, or Russian spring - summer encephalitis (RSSE) subtype. The latter consists of several subtypes prevalent worldwide, such as Louping ill in Scottland, Ireland, and Norway, Omsk hemorraghic fever in Siberia, Kyasanur forest disease in India, and Powassan in North America (Murphy et al. 1995). TBE has been known in Europe since the 1930s. Still rare, but severe and lethal, TBE is an increasing problem. There are indications that a tick-borne encephalitis-like virus has established itself in ticks in the United States (Telford et al. 1997).
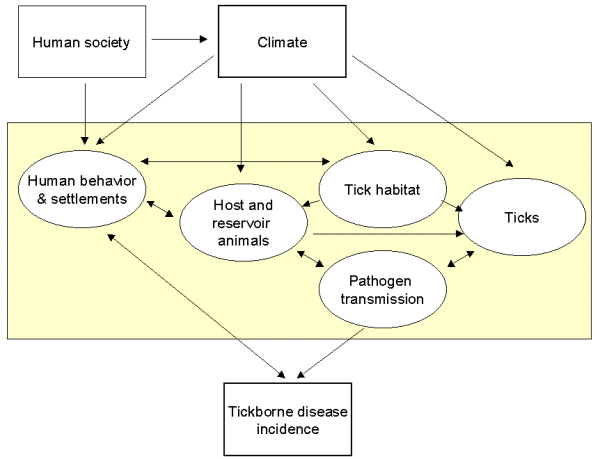
Changes in the macro- and microclimate may influence transmission of Lyme disease and TBE through impact on the life cycle dynamics of the tick, the tick habitat, and the host animals, as well as through changes in human behavior and human-vector-host animal interactions (Cederlund 1981, Dobson and Carper 1993, Jaenson et al. 1994, Kaiser 1995, Lindsay et al. 1995) (Fig. 1).
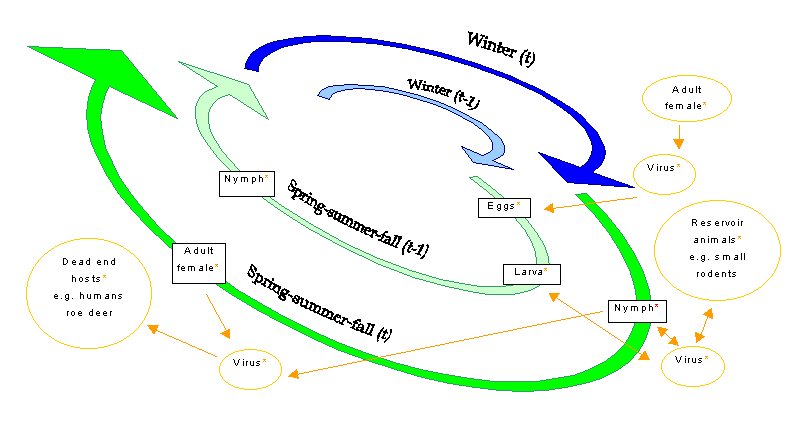
The north-European vector for both Lyme disease and TBE is Ixodes ricinus, a close relative of the North American carrier of Lyme disease, Ixodes scapularis (also known as Ixodes dammini) (Jaenson et al. 1994). The life cycle of I. ricinus stretches over 2 - 3 yr, on average, but may be prolonged up to 6 yr, in some cases, in cold regions where ticks may hibernate even during some spring - summer - fall periods (Balashov 1972, Real 1996). Three out of four development stages, larva, nymph, and adult tick, are blood-sucking. The pathogens can be transmitted transovarially, although the larvae are rarely infected before the first blood meal (Ostfeldt 1997) (Fig. 2).
The northern distribution limit for ticks in Europe lies within the Swedish border. Sweden is situated between latitude 55ºN and 69ºN. Because of the warm North Atlantic Current, the climate in the tick-borne disease-infested parts of the country resembles the climate in states like Washington, Minnesota, and Connecticut in the United States.
Sweden has had an excellent national registration of TBE cases since the late 1950s. According to McMichael et al. (1996), empirical studies on relations between natural fluctuations in contemporary weather and disease incidence are needed to provide basic data for analogue estimates of future impacts of a climate change on tick-borne disease transmission. This study analyzes possible relationships between seasonal variation in temperature and precipitation and changes in TBE incidence in a high-endemic region in Sweden. The study is based on daily minimum and maximum temperatures, monthly precipitation, and disease incidence data during a 36-yr period. Temperature and precipitation variables were adjusted to the length of the tick's life cycle and to bioclimatic thresholds of importance for disease transmission. Multiple regressions were used for the statistical analyses. This is probably the first time that reliable incidence data of a tick-borne disease have been related to meteorological data over several decades. Based on the results, possible reasons behind the correlation between climate and TBE are discussed. We conclude that it is most likely that a temperature increase will exacerbate the incidence and will extend the season of the vector-borne disease, TBE.
Incidence data and study region
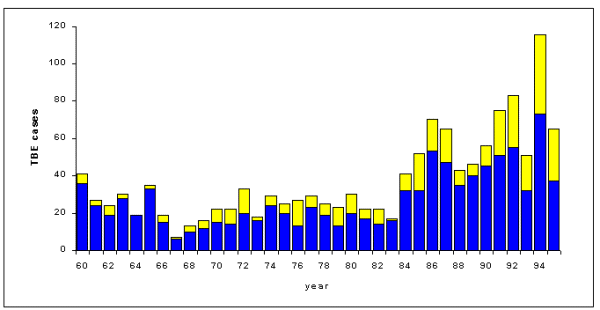
TBE requires higher tick population abundance than does Lyme disease for successful pathogen transmission to humans. In the endemic region, 1 - 4% or less of the ticks are infected with TBE, whereas the same rate for Lyme disease is 3 - 23% (WHO meeting 1986, Gustafson 1994). Only one tick bite out of 600 has been shown to produce clinical symptoms in high-endemic TBE regions, in contrast to one out of 150 bites for Lyme disease (Gustafson et al. 1990, 1992). Therefore, TBE has a more restricted geographical distribution than Lyme disease in Sweden (Holmgren and Forsgren 1990, Berglund et al. 1995), with the highest abundance within Stockholm County (Fig. 3).
An epidemic of TBE occurred in 1956 in Stockholm County. Since then, all hospitalized cases of encephalitis in the region have been serologically tested for TBE virus. Verified TBE cases have been registered at the Swedish Institute for Infectious Disease Control (Holmgren and Forsgren 1990). This study consists of all cases of TBE in Stockholm County from 1960 to 1996. The human population in the region has increased from 1.2 to 1.7 x 106 during the study period. The incidence was computed as the number of cases per 10,000 inhabitants per year.
Meteorological data
Slight variations in local climate exist within the study region. After comparing data from three different meteorological stations, we found the Stockholm Meteorological Station (SMS) to be representative of the regional average. From the SMS, we obtained 24-h measurements of daily minimum temperature for the period October 1958 to January 1996, and monthly precipitation data and daily maximum temperatures for the period 1959 to 1995. To adjust for the tick's life length, each annual TBE incidence was related to two years of temperature and precipitation data, including the year of the TBE value (year t) and the year prior to it (year t -1). Temperature variables were expressed as number of days per year with temperatures above, below, or in between different temperature limits.
Vector-borne disease transmission is only possible within the lower and higher bioclimatic temperature thresholds for vector and pathogen activity (Dobson and Carper 1993, McMichael et al. 1996). The lower temperature thresholds are of more importance than the upper limits for tick-transmitted diseases at high northern latitudes. Lower bioclimatic thresholds of interest for pathogen transmission are daily minimum temperatures that allow tick activity and metamorphosis during the spring - summer - fall season (Fig. 2).
Based on previous tick studies, the following temperature limits were chosen for the analyses: 5º, 8º, and 10ºC (Balashov 1972, McEnroe 1977,1984, Daniel 1993, Fujimoto 1994, Clark 1995, Lindsay et al. 1995). Freezing temperatures during the winter season may affect the survival of ticks and reservoir of host animals, with an indirect effect on disease transmission. Tick nymphs and adults, in particular, especially if fed, may resist freezing temperatures well below -7ºC (Smorodintsev 1958, McEnroe 1984, Daniel 1993, Fujimoto 1994, Lindsay et al. 1995). Accordingly, the following freezing temperature limits were analyzed: 0º, -7º, -10º, -12º, and -15ºC.
Precipitation is of indirect interest for pathogen transmission, through impact on the habitat and on tick activity and survival (Knülle and Rudolph 1983, Daniel 1993, Lindsay et al. 1995). Analyses included monthly precipitation during April to October.
Hot Scandinavian summer days promote increased outdoor activities. It has been suggested that such human behavior may result in increased TBE incidence (Kaiser 1995). The Swedish Meteorological and Hydrological Institute, SMHI, defines hot summer days as days with maximum temperatures above 25ºC. This temperature variable was analyzed for the year of possible pathogen transmission, i.e., year t.
Statistics
Data were analyzed using multiple regression with backward selection, using TBE incidence as the dependent variable and meteorological data as the independent variables. These independent variables consisted of: precipitation (in millimeters) during two consecutive spring - summer - fall seasons; number of days with maximum temperature above 25ºC during the incidence year (t); and number of days with minimum temperature in each of five or six intervals during two consecutive years, t and (t-1). For temperatures below the freezing point, year t and (t -1) were represented by corresponding winter seasons. The minimum temperature limits were varied at the upper and lower end in order to investigate whether the results were sensitive to the particular bioclimatic temperature thresholds chosen. The following sets of minimum temperature intervals were analyzed: below -15ºC/-12ºC/-10ºC/-7ºC; between -15ºC/-12ºC/-10ºC and -7ºC; -7ºC to 0ºC; 0ºC to 5ºC; between 5ºC and 8ºC/10ºC; and above 8ºC/10ºC. Accordingly, either 13 or 15 independent variables were used in the analyses. Eight different multiple regressions were performed, including 116 regression analyses in all.
Regressions with forward selection, and nonparametric correlations and tests (Pearson and Spearman) were also performed.
The different multiple regressions all showed that the combination of a milder winter prior to the incidence year (t -1) and two successive years with milder spring and fall seasons was correlated with increases in TBE incidence.
The winter seasons (t -1) were related to increases in TBE incidence for all of the different sets of temperature intervals below -7ºC. No major difference in correlation strength was found when the temperature limits -7º, -10º, -12º, or -15ºC were used. Temperatures below the freezing point in the interval 0º to -7ºC were not related to TBE incidence. No consistent relations were found between any temperatures below the freezing point for the winter season of the incidence year, t.
Increases in TBE incidence were also related to a greater number of days with minimum temperatures above the threshold temperatures for tick activity and metamorphosis (i.e., with 5ºC, 8ºC, 10ºC as the limits) in both year t and (t -1). The variables expressing number of days with minimum temperatures in the intervals above 5ºC were all correlated with each other.
No consistent relation between precipitation and TBE incidence was found in this study. None of the analyses indicated a relation between TBE incidence and warm summer days, represented by number of days with a maximum temperature above 25ºC.
A representative regression analysis for four variables is shown in Table 1. In going from 15 to four explanatory variables, there was a reduction from R2 = 0.55 to R2 = 0.48, with a considerably steeper reduction as more variables were eliminated. The normal distribution assumption was checked by analyzing the residuals, the Q-Q test giving r = 0.994 (n = 36, P >0.25).
Results of the multiple regression with forward selection, as well as the nonparametric correlations and tests, confirmed these relations, but are not presented in depth here.
|
TABLE 1. Results of regression analysis, which well represent the consistent findings in different multiple regressions of this study. In this regression, the number of days during the winter (t-1) with minimum temperatures between -12º and -7ºC is shown to be significant. The b value is negative for this variable, indicating a relation between a mild winter in (t-1) and an increase in TBE incidence in the year t. The number of days with temperatures below -12ºC was closely correlated with the number of days with temperatures between -12º and -7ºC. For both year t and (t-1), the best correlations with TBE incidence were obtained with 5º -10ºC as the critical interval.
*T = B/Standard error of B; abbreviations used in SPSS for Windows | ||||||||||||||||||||||||||||||||||||||||||||||||
The results showed that milder winters, defined as winters with fewer days with minimum temperatures below -7ºC, were related to increases in TBE incidence a year later. An early arrival of spring and a prolonged fall season during a 2-yr period, represented by an increased number of days per year with minimum temperatures in the intervals above 5ºC, were also shown to enhance the incidence of disease.
It has been projected, and has already been observed in the United States, that, with a shift toward a milder climate, daily minimum temperatures will rise proportionally higher than mean and maximum temperatures (Karl et al. 1993). Results of this study indicate that an increase in seasonal daily minimum temperatures will increase the transmission and extend the season of high-latitude tick-borne diseases.
Regional climate change models estimate a proportionally higher temperature increase during winter at the high northern latitudes (Maskell et al. 1993, IPCC 1996a). Drawing on the findings of this study, it is likely that milder winters will lead to increases in the abundance of TBE in these regions.
No consistent relation was found between precipitation and TBE incidence. Monthly seasonal precipitation is probably too rough an estimate to reflect both the micro- and macroclimate of interest for tick-transmitted diseases.
The strength of the correlations detected may be underestimated. Hundreds of thousands of people living or working outdoors in high-risk locations within Stockholm County have been vaccinated against TBE since a vaccine became available in 1989. Preliminary Austrian data show a 95% effect after adequate initial inoculations, with a need for booster doses every third to fifth year (Immuno AG, Wien). We did not adjust for this in the regression analyses. Neither were snow conditions included, due to great local variations in time and space within the study region. Snow conditions may directly and indirectly affect the vector abundance and, hence, disease incidence, both positively and negatively. Snow cover may partly protect ticks, small host animals, and vegetation, whereas deep snow conditions may be fatal to larger host animals such as roe deer and, in particular, their offspring (Berry 1981, Cederlund 1981, McEnroe 1990).
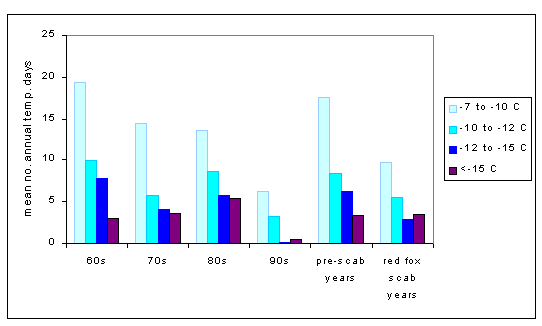
During the last 13 yr of the study period, a decline in the population abundance of red fox, a major predator of roe deer kids, occurred due to scabies infestation. Simultaneously, winters became considerably milder (Fig. 4). This combination led to a rapid increase in roe deer population density. The roe deer is important for adult ticks and their reproduction, but does not have a direct function in transmitting the TBE virus (Fig. 2). Roe deer population data are not available, but statistics on regional hunting limits show a continuous increase in the number of shot animals, up to a peak in 1994 of three times the 1982 value (Swedish Hunting Association, Box 1, 163 21 Spanga, Sweden). During normal conditions, winter starvation is second to hunting as the major cause of death in the Swedish roe deer population, followed by deaths from predation and diseases (Cederlund and Liberg 1995). To investigate how our results were influenced by the red fox scabies (which led to reduced predation on roe deer), we calculated multiple regressions for the period before the rapid decline in red fox populations, 1960 to 1982. The regressions indicated an even stronger correlation between climate and TBE, with some minor variable differences. The coldest winter temperatures for year (t -1) continued to be important. The spring - summer - fall temperatures showed strong relations with TBE for year (t), but not for year (t -1). The very warm summer days, i.e. , >25ºC maximum temperature, were more strongly (negatively) correlated with TBE in 1960 - 1982. Differences compared to the entire study period may be explained by the relatively colder winters during the pre-scabies period (Fig. 4).
Obviously, complex interactions among ticks, host animals such as deer and rodents, habitat availability, and human interference (as discussed by Cederlund 1981, Fruzinski 1983, Anderson and May 1991, Daniel and Kolar 1991, French et al. 1992, Dobson and Carper 1993, Mejlon and Jaenson 1993, Gilot et al. 1994, Jaenson et al. 1994, Buskirk and Ostfeld 1995, Glass et al. 1995, Ostfeld et al. 1995, and Ostfeld 1997), will influence the incidence of TBE. For example, major changes in the functional relationships of ecosystems, such as a decline or increase in host animals, a change in abundance of tick predators, or habitat modifications, may strongly influence tick survival and abundance. In this study, such complex interactions have not been analyzed. They have been treated as a black box. Despite this weakness of the study, significant correlations have been found between climate and TBE, based on detailed data of disease incidence and daily temperatures over a 36-yr period.
Results of this study suggest that a change toward a milder climate may extend the seasonal range of TBE and intensify established endemicity in regions with interspecies relations and climatic conditions similar to those in Sweden.
The impact of seasonal and interannual weather fluctuations on the transmission of tick-borne diseases might differ, however, from effects caused by long-term shifts in background climate. McMichael et al. (1996) suggested that a prolonged climatic change may alter the interspecies balances and critical points may be exceeded. Ecosystem function and resilience may be altered, resulting in a cascade of changes with other bioclimatic thresholds than the present ones.
This paper has focused on changes in TBE incidence from climatic variations within an endemic region. Mathematical prediction models of the impact of climate change on malaria, dengue fever, and schistosomiasis have projected latitude/altitude shifts in the distribution limits (Martens 1995, Martens et al. 1995, 1997). Based on results of the study presented in this paper, one may assume that a change toward a milder climate will also move the geographical distribution limits of TBE northward. The southern distribution limit of the disease will probably also be affected by a change in climate. However, no such assumptions can be made from the results of this study, because the bioclimatic maximum temperature thresholds were not analyzed.
The findings of this study may have implication for other diseases transmitted by the same, or closely related, tick species. Ticks are known to be potent vectors for a whole range of pathogens, including viruses, bacteria, richettsia, and protozoa, which are of importance in both human and veterinary medicine. Lyme disease is already becoming increasingly prevalent in both Europe (Berglund et al. 1995) and the United States. Zoonotic diseases, such as Erlichiosis and Babesiosis, have recently emerged in humans (Dawson et al. 1996), and a TBE-like virus has been detected in deer ticks in the United States (Telford et al. 1997). Thus, a future change in climate may possibly influence the transmission of both present and emerging tick-borne diseases in regions located at high latitudes and altitudes.
Responses to this article are invited. If accepted for publication, your response will be hyperlinked to the article. To submit a comment, follow this link. To read comments already accepted, follow this link.
Acknowledgments
This study was funded by the Swedish Council for Planning and Coordination of Research, the OK Petroleum Environmental Foundation, and the Center for Research on Natural Resources and the Environment, Stockholm University. I would like to thank Dr. Paul R. Epstein of the Harvard Medical School, Boston, United States, for inspiring ideas; Dr Rolf Gustafson of Karolinska Institute at Huddinge University Hospital for support and for making TBE data available; Professor Thomas Polfeldt of Stockholm University, for valuable statistical help; and Prof. Carl Folke, in particular, and Prof. Erik Arrhenius, Prof. Göran Pershagen, and Dr. Lars Tälleklint for constructive comments on the manuscript.
Anderson, R. M., and R. M. May. 1991. Infectious diseases of humans. Dynamics and control. Oxford University Press, UK.
Balashov, Y. S. 1972. Bloodsucking ticks (Ixodoidea), vectors of disease of man and animals. Miscellaneous Publications of the Entomological Society of America 8: 163-376.
Berglund, J., R. Eitrem, K. Ornstein, A. Lindberg, A. Rigner, H. Elmrud, M. Carlsson, A. Runehagen, C.Svanborg, and R. Norrby. 1995. An epidemiological study of Lyme disease in southern Sweden. New England Journal of Medicine 333:1319-1327.
Berry, M. O. 1981. Snow and climate. Pages 51-60 in D. M. Gray, and D. H. Male, editors. The handbook of snow. Principles, processes, management and use. Pergamon, Toronto, Canada.
Buskirk, J. V., and R. S. Ostfeld. 1995. Controlling Lyme disease by modifying the density and species composition of tick hosts. Ecological Applications 5(4): 1133-1140.
Cederlund, G. 1981. Some aspects of roe-deer winter ecology. Dissertation. Stockholm University, Stockholm, Sweden.
Cederlund, G., and O. Liberg. 1995. The roe deer. Wildlife, ecology and hunting. In Swedish (Rådjuret. Viltet, ekologin och jakten). Almqvist and Wiksell, Uppsala, Sweden.
Clark, D. D. 1995. Lower temperature limits for activity of several Ixodid ticks (Acari:Ixodidae): Effects of body size and rate of temperature change. Journal of Medical Entomology 32(4): 449-452.
Daniel, M. 1993. Influence of the microclimate on the vertical distribution of the tick Ixodes ricinus (L.) in central Europe. Acarologica XXXIV: 105-113.
Daniel, M., and J. Kolar. 1991. Using satellite data to forecast the occurrence of the common tick Ixodes ricinus (L.) Modern Acarology 1: 191-196.
Dawson, J. E., C. K. Warner, S. Standaert, and J. G. Olson. 1996. The interface between research and the diagnosis of an emerging tick-borne disease, human Ehrlichiosis due to Ehrlichia chaffeensis. Archives of Internal Medicine 156: 137-142.
Dobson, A., and R. Carper. 1993. Biodiversity. Lancet 342: 1096-1099.
French, J. B., W. L. Schell, J. J. Kazmierczak, and J. P. Davis. 1992. Changes in population density and distribution of Ixodes dammini (Acari: Ixodidae) in Wisconsin during the 1980s. Journal of Medical Entomology 29(5): 723-728.
Fruzinski, B. 1983. Habitat, density and spatial structure of the forest roe deer population. Acta Theriologica 28: 243-258.
Fujimoto, K. 1994. Comparison of the cold hardiness of Ixodes nipponensis and I. persulcatus (Acari: Ixodidae) in relation to the distribution patterns of both species in Chichibu Mountains. Japanese Journal of Sanitary Zoology (Eisei Dobutsus) 1: 333-339.
Gilot, B., C. Guiguen, and B. Degeilh. 1994. Phytoecological mapping of Ixodes ricinus as an approach to the distribution of Lyme borrelisosis in France. Pages 105-112 in J. S. Axford and D. H. E. Rees, editors. Lyme borreliosis. Plenum Press, New York, New York, USA.
Glass, G. E., B. S. Schwartz, J. M. Morgan III, D. T. Johnson, P. M. Noy, and E. Israel. 1995. Environmental risk factors for Lyme disease identified with Geographic Information Systems. American Journal of Public Health 85(7): 944-948.
Gustafson, R., B. Svenungsson, A. Gardulf, G. Stiernstedt, and M. Forsgren. 1990. Prevalence of tick-borne encephalitis and Lyme borreliosis in a defined Swedish population. Scandinavian Journal of Infectious Diseases 22: 297-306.
Gustafson, R., B. Svenungsson, M. Forsgren, A. Gardulf, and M. Granström. 1992. Two-year survey of the incidence of Lyme borreliosis and tick-borne encephalitis in a high-risk population in Sweden. European Journal of Clinical Microbiological Infectious Diseases II: 894-900.
Gustafson, R. 1994. Epidemiological studies of Lyme borreliosis and tick-borne encephalitis. Scandinavian Journal of Infectious Diseases Supplement 24: 181-188.
Hadley Center. 1995. Modelling climate change 1860-2050. The U.K. Meteorological Office, Bracknell, UK.
Holmgren, E. B., and M. Forsgren. 1990. Epidemiology of tick-borne encephalitis in Sweden 1956-1989: A study of 1116 cases. Scandinavian Journal of Infectious Diseases 22: 287-295.
IPCC. Watson, R. T., M. C. Zinyowera, and R. H. Moss, editors. 1996a. Climate change 1995: Impacts, adaptations and mitigation of climate change. Scientific-technical analyses. Contribution of working group II to the Second Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge, UK.
IPCC. Houghton, J. T., L. G. Meira Filho, B. A. Callander, N. Harris, A. Kattenberg, and K. Maskell, editors. 1996b. Climate change 1995: The science of climate change. Contribution of working group I to the Second Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge, UK.
Jaenson, T. G. T., L. Tälleklint, L. Lundqvist, B. Olsen, L. Chirico, and H. Mejlon. 1994. Geographical distribution, host associations, and vector roles of ticks (Acari: Ixodidae, Argasidae) in Sweden. Journal of Medical Entomology 31: 240-256.
Kaiser, R. 1995. Tick-borne encephalitis in southern Germany. Lancet 345: 463.
Karl, T. R., P. D. Jones, and R. W. Knight. 1993. A new perspective on global warming: asymmetric trends of daily maximum and minimum temperatures. Bulletin of the American Meteorological Society 74: 1007-1023.
Knülle, W., and D. Rudolph. 1983. Humidity relationships and water balance of ticks. Pages 34-70 in F. D. Obenchain and R. Galun, editors. Physiology of ticks. Pergamon Press, Oxford, UK.
Levins, R., T. Awerbuch, U. Brinkman, I. Eckardt, P. Epstein, N. Makhoul, C. Albuquerque de Possas, C. Puccia, A. Spielman, and M. E. Wilson. 1994. The emergence of new diseases. American Scientist 2: 52-60.
Lindsay, I. R., I. K. Barker, G. A. Surgeoner, S. A. McEwen, T. J. Gillespie, and J. T. Robinson. 1995. Survival and development of Ixodes scapularisnbsp; (Acari: Ixodidae) under various climatic conditions in Ontario, Canada. Journal of Medical Entomology 32: 143-152.
Martens, W. J. M. 1995. Modelling the effect of global warming on the prevalence of schistosomiasis. The Netherlands: National Institute of Public Health and Environmental Protection, Report Number 461502010, Bithoven, The Netherlands.
Martens, W. J. M., L. W. Niessen, J. Rotmans, T. H. Jetten, and A. J. McMichael. 1995. Potential impact of global climate change on malaria risk. Environmental Health Perspective 103: 458-464.
Martens, W. J. M., T. H. Jetten, and D. A. Ficks. 1997. Sensitivity of malaria, schistosomiasis and dengue to global warming. Climatic Change 32(2): 145-156.
Martin, P. H., and M. G. Lefebvre. 1995. Malaria and climate: sensitivity of malaria potential transmission to climate. AMBIO XXIV: 200-209.
Maskell, K., I. M. Mintzer, and B. A. Callander. 1993. Basic science of climate change. Lancet 342: 1027-1031.
Matsuoka, Y., and K. Kai. 1994. An estimation of climate change effects on malaria. Journal of Global Environment Engineering 1: 1-15.
McEnroe, W. D. 1977. The restriction of the species range of Ixodes scapularis, Say, in Massachussets by fall and winter temperature. Acarologia XVIII: 618-625.
_______ . 1984. Winter survival and spring breeding by the fall tick, Ixodes dammini, in Massachusetts (Acarina: Ixodidae). Acarologica XXV: 223-229.
_______ . 1990. Climatic regulation of the two-cohort population of Ixodes dammini in coastal Massachusetts. Acarologica XXXI: 235-239.
McMichael, A. J., A. Haines, R. Sloff, and S. Kovats. 1996. Climate change and human health. World Health Organization, Geneva, Switzerland.
Mejlon, H. A., and T. G. T Jaenson. 1993. Seasonal prevalence of Borrelia burgdorferi inIxodes ricinus in different vegetation types in Sweden. Scandinavian Journal of Infectious Diseases 25: 449-456.
Murphy, F. A., C. M. Fauquet, D. H. L. Bishop, S. A. Ghabrial, and A. W. Jarvis, editors. 1995. Virus taxonomy. Sixth Report of the International Committee on Taxonomy of Viruses. Springer-Verlag, Wien, Austria.
Ostfeld, R. S. 1997. The ecology of Lyme-disease risk. American Scientist 85: 338-346.
Ostfeld, R. S., O. M. Cepeda, K. R. Hazler, and M. C. Miller. 1995. Ecology of Lyme disease: habitat associations of ticks (Ixodes scapularis ) in a rural landscape. Ecological Applications 5(2): 353-361.
Patz, J. A., P. R. Epstein, T. A. Burke, and J. M. Balbus. 1996. Global climate change and emerging infectious diseases. Journal of the American Medical Association 275: 217-223.
Randolph, S. E., L. Gern, and P. A. Nuttall. 1996. Co-feeding ticks: epidemiological significance for tick-borne pathogen transmission. Parasitology Today 12: 472-479.
Real, L. A. 1996. Sustainability and the ecology of infectious disease. BioScience 46: 88-97.
Reeves, W. C., J. L. Hardy, W. K. Reisen, and M. M. Milby. 1994. Potential effect of global warming on mosquito-borne arboviruses. Journal of Medical Entomology 31(3): 323-332.
Smorodintsev, A. 1958. Tick-borne spring - summer encephalitis. Progress in Medical Virology 1: 210-248.
Telford, S. R., P. M. Armstrong, P. Katavolus, I. Foppa, A. S. Olmeda Garcia, M. L. Wilson, and A. Spielman. 1997. A new tick-borne encephalitis-like virus infecting New England deer ticks, Ixodes dammini. Emerging Infectious Diseases URL: http://www.cdc.gov/ncidod/EID/vol3no2/telford.htm
WHO meeting. 1986. Tick-borne encephalitis and haemorrhagic fever with renal syndrome in Europe: report on a WHO meeting, Baden, Austria, 3-5 October, 1983. EURO Report Studies 104: 1-51.
Address of Correspondent:
Elisabet Lindgren
Natural Resources Management
Department of Systems Ecology
Stockholm University
S-106 91 Stockholm
Sweden
Phone: +46-8-16 12 90
Fax: +46-8-15 84 17
elisa@system.ecology.su.se
*The copyright to this article passed from the Ecological Society of America to the Resilience Alliance on 1 January 2000.