|
|
|
Copyright © 1997 by The Resilience Alliance*
Ludwig, D., B. Walker, and C. S. Holling. 1997. Sustainability, stability, and resilience. Conservation Ecology [online]1(1): 7. Available from the Internet. URL: http://www.consecol.org/vol1/iss1/art7/
A version of this article in which text, figures, tables, and appendices are separate files may be found by following this link.
Insight Sustainability, Stability, and Resilience Donald Ludwig1, Brian Walker2, and Crawford S. Holling3
1Department of Mathematics, University of British Columbia; 2Division of Wildlife and Ecology, CSIRO; 3Department of Zoology, University of Florida
- Abstract
- Introduction
- A Simple Prototype
- Hard Loss of Stability and Hysteresis
- A Competitive Grazing System
- Fire in a Savanna System
- Concluding Remarks
- Responses
- Literature Cited
KEY WORDS: bifurcation; multiple stable states; resilience; stability.
Equilibrium A mechanical system is at equilibrium if the forces acting on it are in balance. For example, when a body floats, the force of gravity is balanced by the buoyant force due to displacement of the liquid. The "balance of nature'' (Pimm 1991) is an extension of this idea to the natural world. The concept usually refers to steady flows of energy and materials, rather than to a system whose components do not change.
Resilience and stability We are interested in characterizing natural systems that are resilient, i.e., that tend to maintain their integrity when subject to disturbance (Holling 1973). This is related to the idea of stability. The informal concept of stability refers to the tendency of a system to return to a position of equilibrium when disturbed. If a weight is added suddenly to a raft floating on water, the usual response is for the weighted raft to oscillate, but the oscillations gradually decrease in amplitude as the energy of the oscillations is dissipated in waves and, eventually, in heat. The weighted raft will come to rest in a different position than the unweighted raft, but we think of the new configuration as essentially the same as the old one. The system is stable.
If we gradually increase the weight on the raft, eventually the configuration will change. If the weight is hung below the raft, the raft will sink deeper and deeper into the water as more and more displacement is required to balance the higher gravitational force. Eventually, the buoyant force cannot balance the gravitational force and the whole configuration sinks: the system is no longer stable. On the other hand, if the weight is placed on top of the raft, the raft may flip over suddenly and lose the weight and its other contents long before the point at which the system, as a whole, would sink. This sudden loss of stability may be more dangerous than the gradual sinking, because there may be little warning or opportunity to prepare for it. We may think of the raft system as losing its resilience as more weight is placed on it.
Suppose that we accept the ``balance of nature'' and the steady flows of resources that it implies. As we demand more and more of the products of natural systems, and we load them with more and more of our waste products, are we likely to experience a gradual loss of stability or a sudden one? In order to clarify such questions, we must refine our terminology. To decide whether a system is stable or not, we must first specify what we mean by a change in configuration or loss of integrity. If we don't care whether the raft flips over when weighted, then there is no problem of sudden loss of stability for the floating raft. We must also specify the types and quantities of disturbances that may affect the system. Suppose that a fixed weight is placed on top of an occupied raft. If the occupants of the raft move about, the raft may float at a slightly different angle, but if they move too far or all at once, the raft may tip. The range of possible movements of the occupants that do not lead to tipping is called the domain of stability, or domain of attraction, of the upright state. If the amount of the fixed weight is gradually increased, the balance becomes more precarious and, hence, the domain of attraction will shrink. Eventually, the weight becomes large enough so that there is no domain of attraction at all, and the raft will flip over no matter what its occupants do.
The preceding example makes a distinction between the weight loading the raft and the positions of the occupants. If the amount of the weight changes very slowly or not at all, we may think of the "system" as consisting of the raft and weight. The occupants change position relatively quickly, and these changes may be thought of as disturbances of the system. On the other hand, we may adopt a more comprehensive point of view, seeing the raft, the weight, and the occupants as a single system. If the occupants organize themselves to anticipate and correct for external disturbances, then the system may be able to maintain its integrity long enough for them to achieve their objectives. Another possible response to disturbance might be to restructure the raft itself. If it were constructed of several loosely coupled subunits, then excessive weighting or a strong disturbance might flip one part of the system, but leave the rest intact. Such a structure might not require as much vigilance to maintain as the single raft, and it might be able to withstand a greater variety of external disturbances. On the other hand, if the bindings that link the subunits become stiff, then the structure may become brittle and, hence, more prone to failure. This simple example illustrates how the notion of resilience of a system depends upon our objectives, the time scale of interest, the character and magnitude of disturbances, the underlying structure of the system, and the sort of control measures that are feasible.
Section 2 presents the main ideas of stability and resilience for simple, one-dimensional prototype systems. Calculations can be done explicitly for these prototype systems, but their qualitative behavior holds for much more complicated examples. Section 3 illustrates the ideas of bistable equilibria, hard loss of stability, hysteresis, and resilience with a model for the spruce budworm. There are qualitative similarities between the behavior of the budworm model and a variety of ecological systems, particularly lake ecosystems, the Baltic Sea, and boreal forests, although no attempt is made here to provide a formal model for these systems. Such a model is given in Section 4 for a competitive grazing system. This system has qualitative behavior analogous to a one-dimensional model, and it also exhibits hysteresis and hard loss of stability. An analogous system that involves fire as a regulating process is presented in Section 5. The latter system also exhibits regular oscillations. The Appendix presents a detailed account of the relationship between return times for a disturbed system and its resilience. There are two conflicting definitions of resilience, which may cause confusion. The definition of Pimm (1991) applies only to behavior of a linear system, or behavior of a nonlinear system in the immediate vicinity of a stable equilibrium where a linear approximation is valid. For Pimm, loss of resilience is due to slow dynamics near a stable equilibrium. The definition of Holling (1973) that we use here refers to behavior of a nonlinear system near the boundary of a domain of attraction. Loss of resilience, in our sense, is associated with slow dynamics in a region that separates domains of attraction.
Global stability The concept of the balance of nature might be taken to imply that the system will maintain its integrity under any sort of perturbation. Such an assumption may be made (often unconsciously) when we make large modifications to natural systems. Our expectation is that things will proceed more or less as before, and that the response of the system will be approximately proportional to the perturbation. Such behavior is shown by the simplest linear models. Some might argue that a principle of parsimony dictates that such models be used in the absence of strong evidence to the contrary. The following linear model illustrates the property of global stability, which implies that the system will always return to a certain equilibrium, regardless of how far it is displaced from that equilibrium.
Suppose that the dynamics are given by a relation of the form

where 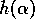 is a
smoothly varying function of an external variable
is a
smoothly varying function of an external variable 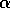 and
and  is the quantity of interest. Then
is the quantity of interest. Then
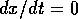 if
if  ; the system has a
single equilibrium there. This equilibrium is stable, since
; the system has a
single equilibrium there. This equilibrium is stable, since 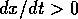 if
if 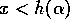 , and
, and 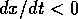 if
if 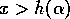 . These relations imply that the
system approaches the equilibrium, no matter what the starting point.
. These relations imply that the
system approaches the equilibrium, no matter what the starting point.
A system such as (Eq. 1) cannot fail us or surprise us. It returns to
an equilibrium, no matter how far it is displaced, and the position of
the equilibrium changes smoothly with the exogenous variable 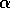 . Such a system is
not suitable for a discussion of possible collapses of natural
systems, since such collapses are excluded by assumptions such as
(Eq. 1). Mathematical theory provides numerous examples of different
behavior, and our goal is to investigate their
plausibility. Unfortunately much theory (including most economic
theory) has been based upon assumptions analogous to (Eq. 1). In
particular, ``resilience'' has been defined by Pimm (1991) in terms of
the system (Eq. 1), and this very special assumption may mislead the
unwary. There is a mathematical theory that shows that systems such as
(1) are good approximations to general systems with a stable
equilibrium, but that theory implies that the approximation holds only
in the immediate vicinity of the equilibrium, i.e., that the
approximation is valid only locally. Details are provided in the
Appendix.
. Such a system is
not suitable for a discussion of possible collapses of natural
systems, since such collapses are excluded by assumptions such as
(Eq. 1). Mathematical theory provides numerous examples of different
behavior, and our goal is to investigate their
plausibility. Unfortunately much theory (including most economic
theory) has been based upon assumptions analogous to (Eq. 1). In
particular, ``resilience'' has been defined by Pimm (1991) in terms of
the system (Eq. 1), and this very special assumption may mislead the
unwary. There is a mathematical theory that shows that systems such as
(1) are good approximations to general systems with a stable
equilibrium, but that theory implies that the approximation holds only
in the immediate vicinity of the equilibrium, i.e., that the
approximation is valid only locally. Details are provided in the
Appendix.
Bifurcation In order to explore the differences between local and global stability, we must examine nonlinear models, i.e., models in which the state variable appears in more complicated functions than linear ones. The following example has three equilibria instead of a single one. Such a complication requires a cubic or more compliocated dependence upon the
 variable, for example,
variable, for example,

Here,
 is a
parameter or a slowly varying quantity whose dynamics are not of
immediate concern. The equilibria of the system are the states where
is a
parameter or a slowly varying quantity whose dynamics are not of
immediate concern. The equilibria of the system are the states where
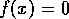 . These are the
states where either of the two factors in (Eq. 2) vanishes. Hence,
they are points where
. These are the
states where either of the two factors in (Eq. 2) vanishes. Hence,
they are points where

If
 , then there are three equilibria
, then there are three equilibria
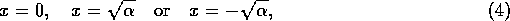
but if
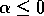 , then there is only the single equilibrium at
, then there is only the single equilibrium at 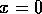 . Such a change in the configuration or stability of equilibria is a called a ``bifurcation'' (Guckenheimer and Holmes 1983). It implies a change in the qualitative behavior of the system. To explore this feature, we must discuss some additional concepts.
. Such a change in the configuration or stability of equilibria is a called a ``bifurcation'' (Guckenheimer and Holmes 1983). It implies a change in the qualitative behavior of the system. To explore this feature, we must discuss some additional concepts.
Local stability and domain of attraction In order to determine the stability of equilibria, it suffices to examine the sign of the velocity of
 . For example, if
. For example, if 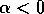 , the second factor in
(Eq. 2) is always positive and, hence,
, the second factor in
(Eq. 2) is always positive and, hence, 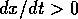 if
if  and
and 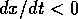 if
if 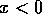 . In this case, the system always
moves away from the state where
. In this case, the system always
moves away from the state where 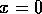 . We conclude that the equilibrium
at
. We conclude that the equilibrium
at 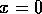 is unstable if
is unstable if
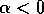 . On the other
hand, if
. On the other
hand, if  , then
, then
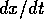 changes sign at
three places:
changes sign at
three places:
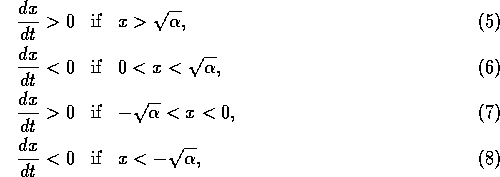
The equilibrium where
 is unstable, because the system
always moves away from that point if nearby (according to Eqs. 5 and
6). Similarly, the equilibrium where
is unstable, because the system
always moves away from that point if nearby (according to Eqs. 5 and
6). Similarly, the equilibrium where 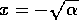 is unstable (according to Eqs. 7
and 8). On the other hand, the equilibrium where
is unstable (according to Eqs. 7
and 8). On the other hand, the equilibrium where 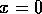 is stable (according to Eqs. 6 and
7), because the motion from nearby points is toward that
point. However, if the system starts outside the interval
is stable (according to Eqs. 6 and
7), because the motion from nearby points is toward that
point. However, if the system starts outside the interval 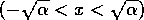 , it moves away from
the equilibrium at
, it moves away from
the equilibrium at 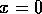 . Therefore, the equilibrium at
. Therefore, the equilibrium at
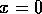 is locally
stable, but not globally stable. The system returns to
is locally
stable, but not globally stable. The system returns to 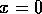 if small perturbations
are made, but larger perturbations take the system into an unstable
domain. The interval
if small perturbations
are made, but larger perturbations take the system into an unstable
domain. The interval 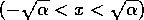 is called the ``domain of
attraction'' of the point
is called the ``domain of
attraction'' of the point 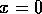 , because trajectories that start
within that interval eventually return to
, because trajectories that start
within that interval eventually return to 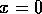 , but not those that start outside.
, but not those that start outside.
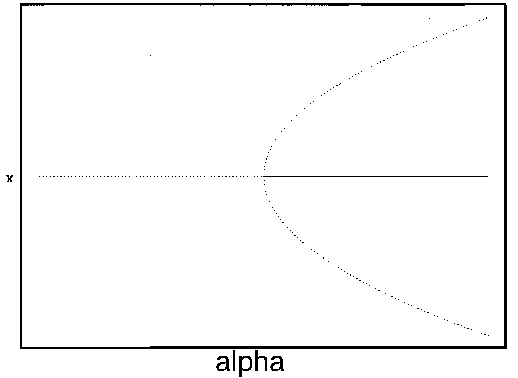
It is clear that the domain of attraction of the stable equilibrium at
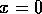 shrinks as
shrinks as  decreases toward
zero. The three equilibria collapse into one where
decreases toward
zero. The three equilibria collapse into one where 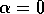 , and only a single unstable
equilibrium remains when
, and only a single unstable
equilibrium remains when  . This information is summarized
in Fig. 1
. The diagram looks like a branch and, for this reason, it is
called a ``bifurcation diagram." The domain of attraction of the point
. This information is summarized
in Fig. 1
. The diagram looks like a branch and, for this reason, it is
called a ``bifurcation diagram." The domain of attraction of the point
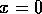 is contained
within the two curved branches, and there is no other domain of
attraction.
is contained
within the two curved branches, and there is no other domain of
attraction.
FIG. 1. The parameter 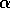 is plotted on the horizontal axis and the corresponding equilibria in
is plotted on the horizontal axis and the corresponding equilibria in  for Eq. (2)are plotted on the vertical axis. The stable equilibrium is plotted with a solid curve, whereas unstable ones are plotted with dotted curves.
for Eq. (2)are plotted on the vertical axis. The stable equilibrium is plotted with a solid curve, whereas unstable ones are plotted with dotted curves.
Increasing evidence has accumulated for the existence of multistable
states in nature: coral reefs (Done 1992, Hughes 1994, McClanahan et
al. 1996), African rangelands (Dublin et al. 1990), shallow lakes
(Schindler 1990, Carpenter and Leavitt 1991, Scheffer et al. 1993),
kelp forests (Estes and Duggins 1995), and grasslands (D'Antonio and
Vitousek 1992, Zimov et al. 1995).
Disturbances and slow parameter changes
We have seen that if , then this system approaches the
stable equilibrium at
, then this system approaches the
stable equilibrium at 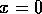 if it is started within the domain
of attraction. If we envisage disturbances that displace the system a
distance
if it is started within the domain
of attraction. If we envisage disturbances that displace the system a
distance 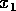 from the
stable equilibrium, they will not affect the integrity of the system
(its tendency to return to the 0 state) as long as
from the
stable equilibrium, they will not affect the integrity of the system
(its tendency to return to the 0 state) as long as 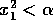 . Now, if we allow the parameter
. Now, if we allow the parameter
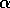 to decrease
slowly toward
to decrease
slowly toward  ,
the system will take longer and longer to return to the state
,
the system will take longer and longer to return to the state 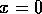 when
when  is displaced to
is displaced to 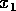 , because motion is very slow near
, because motion is very slow near
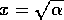 , and a
disturbance of magnitude
, and a
disturbance of magnitude 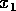 may take the system into the region
of slow dynamics. We may think of the decrease in
may take the system into the region
of slow dynamics. We may think of the decrease in  as causing a loss of resilience,
because the integrity of the system is threatened more and more by
disturbances of a given magnitude. A symptom of loss of resilience may
be longer and longer times to return to the vicinity of
as causing a loss of resilience,
because the integrity of the system is threatened more and more by
disturbances of a given magnitude. A symptom of loss of resilience may
be longer and longer times to return to the vicinity of 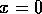 after disturbance. The
connection between return times and resilience is not completely
straightforward; we address it in some detail in the Appendix.
after disturbance. The
connection between return times and resilience is not completely
straightforward; we address it in some detail in the Appendix.
Two domains of attraction The preceding system is not a believable model for natural systems, because it predicts that the state variable may approach infinity under some circumstances. A more plausible scenario is one in which the system may change from having a single stable equilibrium to one with two stable equilibria. We next consider a number of such ``bistable'' systems.
We obtain a simple prototype for such systems by changing the sign of
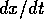 in (Eq. 2). If
the direction of time is reversed, the stable and unstable equilibria
are interchanged. If
in (Eq. 2). If
the direction of time is reversed, the stable and unstable equilibria
are interchanged. If 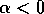 , then the single equilibrium at
, then the single equilibrium at
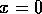 is globally
stable: the system always returns to that equilibrium no matter where
it starts. Instead of two unstable equilibria when
is globally
stable: the system always returns to that equilibrium no matter where
it starts. Instead of two unstable equilibria when  (as in the former case), there will
be two stable equilibria. The new system is
(as in the former case), there will
be two stable equilibria. The new system is
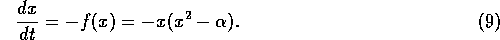
If  , for this system, we have
, for this system, we have
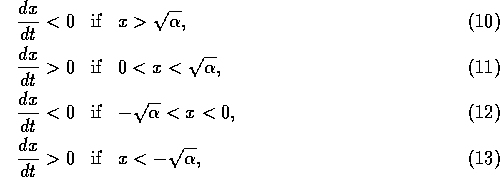
If  initially,
then
initially,
then  heads toward
the equilibrium at
heads toward
the equilibrium at  , but if
, but if 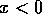 initially, then
initially, then  heads toward the equilibrium at
heads toward the equilibrium at
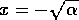 . Thus, the
domain of attraction of the point
. Thus, the
domain of attraction of the point  is the positive
is the positive  axis, and the domain of attraction
of
axis, and the domain of attraction
of 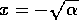 is the
negative
is the
negative  axis. Each of the stable equilibria is locally stable, but not
globally stable. This system can be flipped from one stable state to
another by crossing the unstable line where
axis. Each of the stable equilibria is locally stable, but not
globally stable. This system can be flipped from one stable state to
another by crossing the unstable line where 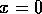 . Because this line separates the
two domains of attraction, it is called a ``separatrix." The
equilibria in
. Because this line separates the
two domains of attraction, it is called a ``separatrix." The
equilibria in  are
plotted in Fig. 2
.
are
plotted in Fig. 2
.
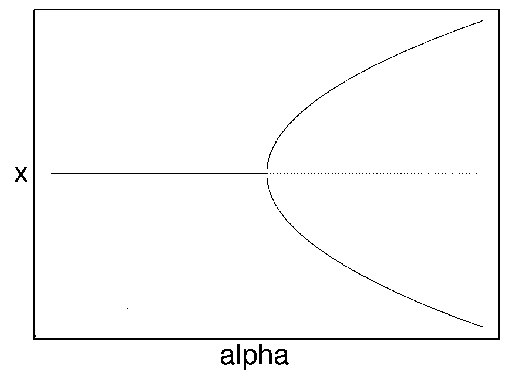
FIG. 2. Equilibria in  for Eq. (9) are plotted vs.
for Eq. (9) are plotted vs. 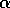 , as in
Fig. 1. The direction of increase of time is reversed as compared with Fig. 1; hence,the stable and unstable equilibria are interchanged.
, as in
Fig. 1. The direction of increase of time is reversed as compared with Fig. 1; hence,the stable and unstable equilibria are interchanged.
Disturbances and slow parameter change
The bifurcation diagram in Fig. 2 implies a great deal about the response of the system to disturbance. If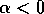 , then the system will return to
the stable equilibrium at
, then the system will return to
the stable equilibrium at 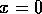 , no matter how large the
disturbance: there is nowhere else for it to go. However, if
, no matter how large the
disturbance: there is nowhere else for it to go. However, if  and the system starts
near the lower branch, it will tend to return there if displaced by a
small amount. As
and the system starts
near the lower branch, it will tend to return there if displaced by a
small amount. As 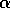 decreases toward zero, the distance between the stable equilibria and
the unstable one decreases. Hence, disturbances of a given magnitude
take the system closer and closer to the unstable
equilibrium. Dynamics are slow near the unstable equilibrium and,
hence, the time to return to the vicinity of the lower branch
increases sharply for trajectories that approach the unstable
equilibrium. This point about return times can be made precise by a
calculation analogous to that in the Appendix.
decreases toward zero, the distance between the stable equilibria and
the unstable one decreases. Hence, disturbances of a given magnitude
take the system closer and closer to the unstable
equilibrium. Dynamics are slow near the unstable equilibrium and,
hence, the time to return to the vicinity of the lower branch
increases sharply for trajectories that approach the unstable
equilibrium. This point about return times can be made precise by a
calculation analogous to that in the Appendix.
For a higher level of disturbance and  , the system may be moved across the
separatrix at
, the system may be moved across the
separatrix at 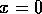 ,
and may approach the upper branch of stable equilibria. Under a random
pattern of disturbances, we may expect to see the system spend long
periods of time in the vicinity of one or the other of the stable
equilibria. Every now and then, the random disturbances may combine
and send the system to the other stable equilibrium. The ``Allee
effect'' studied by ecologists provides an example of a bistable
system. A population may suffer reduced survival or reproductive
success at low numbers. For example, schooling fishes tend to suffer
low per capita mortality if their numbers are high enough relative to
the capacity of their predators. If such fish are reduced in numbers
through fishing pressure or environmental degradation, the population
may decline and eventually become extinct locally. On the other hand,
a large population may sustain itself over long periods. Dynamics of
this sort might explain the occasional flips between dominance of
sardine and anchovy, as revealed by deposits of their scales off the
coast of California. A similar pattern appears in such geophysical
features as the polarity of the earth's magnetic field, the ocean
circulation involving the Gulf Stream, and climate fluctuations, but
some of these fluctuations may be too regular to be completely
random. For larger values of
,
and may approach the upper branch of stable equilibria. Under a random
pattern of disturbances, we may expect to see the system spend long
periods of time in the vicinity of one or the other of the stable
equilibria. Every now and then, the random disturbances may combine
and send the system to the other stable equilibrium. The ``Allee
effect'' studied by ecologists provides an example of a bistable
system. A population may suffer reduced survival or reproductive
success at low numbers. For example, schooling fishes tend to suffer
low per capita mortality if their numbers are high enough relative to
the capacity of their predators. If such fish are reduced in numbers
through fishing pressure or environmental degradation, the population
may decline and eventually become extinct locally. On the other hand,
a large population may sustain itself over long periods. Dynamics of
this sort might explain the occasional flips between dominance of
sardine and anchovy, as revealed by deposits of their scales off the
coast of California. A similar pattern appears in such geophysical
features as the polarity of the earth's magnetic field, the ocean
circulation involving the Gulf Stream, and climate fluctuations, but
some of these fluctuations may be too regular to be completely
random. For larger values of 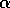 , one would expect flips from one
equilibrium to the other to be extremely rare. Thus, we may associate
an increase in
, one would expect flips from one
equilibrium to the other to be extremely rare. Thus, we may associate
an increase in 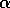 with an increase in resilience.
with an increase in resilience.
The following model was used by Ludwig, Jones, and Holling (1978) to understand the dynamics of the spruce budworm. The quantity 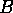 represents budworm density, measured in larvae per acre. This density is assumed to vary in time according to
represents budworm density, measured in larvae per acre. This density is assumed to vary in time according to
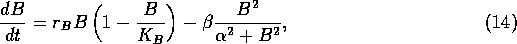
where 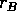 is an intrinsic growth rate at low densities,
is an intrinsic growth rate at low densities, 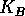 is a carrying capacity for the budworm in the absence of predation, and the second term in (Eq. 14) is a predation rate. The predators are assumed to have a Holling type-III functional response, with a maximum predation rate of
is a carrying capacity for the budworm in the absence of predation, and the second term in (Eq. 14) is a predation rate. The predators are assumed to have a Holling type-III functional response, with a maximum predation rate of 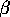 and a half-saturation budworm density of
and a half-saturation budworm density of  . This functional form implies that predators have their greatest influence upon dynamics at intermediate ranges of budworm densities. At low densities, the predators search for alternate prey, because returns from foraging for budworm are relatively low. At high densities, budworms swamp their predators; thus, the predators have a small per capita effect, just as predators have a small per capita effect on large schools of fish. The parameter
. This functional form implies that predators have their greatest influence upon dynamics at intermediate ranges of budworm densities. At low densities, the predators search for alternate prey, because returns from foraging for budworm are relatively low. At high densities, budworms swamp their predators; thus, the predators have a small per capita effect, just as predators have a small per capita effect on large schools of fish. The parameter  is proportional to a measure of foliage density, because the predators search foliage for the budworms and their response is mediated by the number of budworms per unit of foliage. Hence,
is proportional to a measure of foliage density, because the predators search foliage for the budworms and their response is mediated by the number of budworms per unit of foliage. Hence, 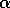 is actually a state variable that generally changes on a
slower time scale than the budworm. For the moment, we regard
is actually a state variable that generally changes on a
slower time scale than the budworm. For the moment, we regard 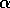 as a constant.
as a constant.
Some algebra supplied in Ludwig et al. (1978) shows that there are
either two or four equilibria for the budworm, depending upon the
sizes of the dimensionless parameters 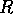 and
and 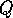 , given by
, given by

These equilibria satisfy
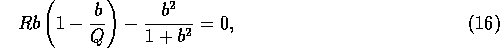
where 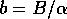 . The
equilibrium
. The
equilibrium 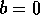 is
always unstable, because
is
always unstable, because 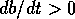 if
if 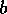 is small and positive. The highest
equilibrium is always stable, because
is small and positive. The highest
equilibrium is always stable, because 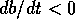 if
if 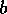 is very large and positive. Thus,
if there are only two equilibria, budworm density always moves toward
the upper equilibrium. When there are four equilibria, they alternate
in stability. A typical case is shown in Fig. 3
. If
is very large and positive. Thus,
if there are only two equilibria, budworm density always moves toward
the upper equilibrium. When there are four equilibria, they alternate
in stability. A typical case is shown in Fig. 3
. If 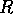 is between
is between 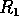 and
and 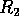 ,
, 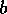 may approach either the high
equilibrium or the low equilibrium, depending upon whether the
starting position of
may approach either the high
equilibrium or the low equilibrium, depending upon whether the
starting position of 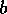 is above or below the unstable
equilibrium, which is the separatrix.
is above or below the unstable
equilibrium, which is the separatrix.
FIG. 3. The equilibria in 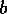 for Eq. (16) are plotted against
for Eq. (16) are plotted against 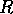 for
for 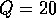 . The points
. The points 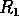 and
and 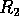 delimit the interval on the
delimit the interval on the 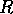 axis where there are three nonzero equilibria for the system.
axis where there are three nonzero equilibria for the system.
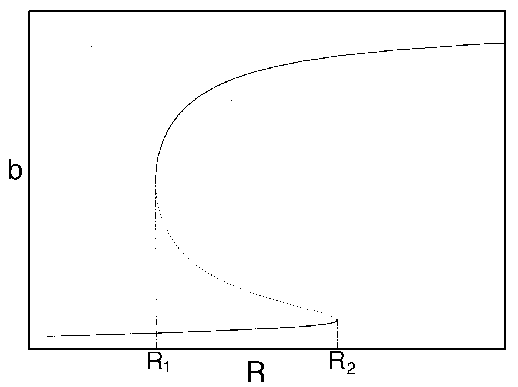
Hard loss of stability Imagine that the parameter
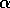 begins at a low value and gradually
increases as the forest grows. It turns out that
begins at a low value and gradually
increases as the forest grows. It turns out that 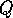 does not change with forest growth;
hence, Fig. 3
applies. Because
does not change with forest growth;
hence, Fig. 3
applies. Because 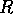 is proportional to
is proportional to 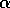 ,
, 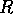 will increase. At first (when
will increase. At first (when 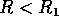 ), budworm numbers
will remain low, as the only stable equilibrium is the low one. Even
when
), budworm numbers
will remain low, as the only stable equilibrium is the low one. Even
when 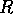 increases
beyond
increases
beyond 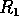 , the
budworm numbers will remain low, because they lie below the unstable
equilibrium, which determines the domain of attraction of the low
equilibrium. The stability of the low equilibrium becomes precarious
as
, the
budworm numbers will remain low, because they lie below the unstable
equilibrium, which determines the domain of attraction of the low
equilibrium. The stability of the low equilibrium becomes precarious
as 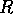 approaches
approaches
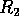 , because the
domain of attraction shrinks. Finally, at
, because the
domain of attraction shrinks. Finally, at 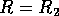 , the lower two equilibria
disappear and budworm density jumps to the high value: an outbreak
occurs. This abrupt change in the attracting state is called a "hard
loss of stability." It should be contrasted with the soft loss of
stability displayed by the system in (Eq. 9). In the case of the
budworm, once density has reached the high equilibrium, there is no
easy way to reduce it to the lower equilibrium. If the variable
, the lower two equilibria
disappear and budworm density jumps to the high value: an outbreak
occurs. This abrupt change in the attracting state is called a "hard
loss of stability." It should be contrasted with the soft loss of
stability displayed by the system in (Eq. 9). In the case of the
budworm, once density has reached the high equilibrium, there is no
easy way to reduce it to the lower equilibrium. If the variable 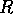 is reduced below
is reduced below 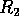 , the budworm remains
at the high equilibrium. As
, the budworm remains
at the high equilibrium. As 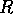 is further reduced, there is a
second hard loss of stability as
is further reduced, there is a
second hard loss of stability as 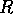 declines below
declines below 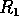 . In this case, there is a jump
down to the low equilibrium, which is not reversed as
. In this case, there is a jump
down to the low equilibrium, which is not reversed as 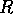 increases again.
increases again.
Hysteresis and cycles If we now connect the dynamics of the trees and the dynamics of the budworm, a new phenomenon appears. If the system starts with low foliage density and low budworm numbers, the foliage density slowly increases until it surpasses
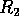 . At this point, an outbreak
occurs, as shown previously. High budworm numbers eventually cause
death of trees, so
. At this point, an outbreak
occurs, as shown previously. High budworm numbers eventually cause
death of trees, so 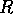 begins to decrease when the budworm
has an outbreak. Budworm numbers remain high even though
begins to decrease when the budworm
has an outbreak. Budworm numbers remain high even though 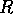 declines, because
budworm density lies above the separatrix. As
declines, because
budworm density lies above the separatrix. As 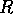 continues to decline to
continues to decline to 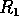 , budworm density
declines slowly and then jumps to a low value when
, budworm density
declines slowly and then jumps to a low value when 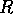 decreases below
decreases below 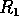 . The different paths followed by
the total system for increasing vs. decreasing
. The different paths followed by
the total system for increasing vs. decreasing 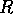 constitute the "hysteresis effect."
The combination of budworm and forest dynamics produces stable cycles
with long periods. Such stable cycles that are maintained through
alternations of rapid transions and slow changes are called
``relaxation oscillations." They are common in many physical,
chemical, and physiological systems (Edelstein-Keshet 1988).
constitute the "hysteresis effect."
The combination of budworm and forest dynamics produces stable cycles
with long periods. Such stable cycles that are maintained through
alternations of rapid transions and slow changes are called
``relaxation oscillations." They are common in many physical,
chemical, and physiological systems (Edelstein-Keshet 1988).
Disturbances and resilience If the objective of management is to keep budworm numbers and foliage damage low, the loss of stability as
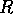 increases beyond
increases beyond 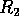 may be regarded as a loss of
resilience. This model suggests that small disturbances near the lower
stable equilibrium may exhibit long return times if they approach the
unstable branch. Such long return times may be a useful diagnostic
indicator. However, because
may be regarded as a loss of
resilience. This model suggests that small disturbances near the lower
stable equilibrium may exhibit long return times if they approach the
unstable branch. Such long return times may be a useful diagnostic
indicator. However, because 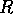 increases as trees grow, a loss of
stability accompanied by a budworm outbreak seems inevitable.
increases as trees grow, a loss of
stability accompanied by a budworm outbreak seems inevitable.
We may adopt a different perspective and regad periodic budworm outbreaks as part of a stable system that renews the forest from time to time. Indeed, systems analogous to the budworm-forest system frequently appear as stable oscillators. The advantage of such oscillators is that they continue to oscillate more or less with the same frequency and amplitude under a wide variety of disturbances. Hence, physiological oscillators are important in maintaining integrity of the organism, which is another kind of resilience. According to this perspective, an attempt to halt the oscillations may lead to a disastrous breakdown in the long term. Will human interventions to increase productivity in natural systems suffer a similar fate?
Lake dynamics Carpenter et al. (1996) have discussed the applicability of these ideas to lake ecosystems. They characterize lake dynamics as either "normal" or "pathological." Normal lakes have high numbers of game fish, effective grazing upon phytoplankton, and low incidence of algal blooms. The normal system maintains its integrity when subjected to perturbations such as phosphorus pulses, because phosphorus moves rapidly into the higher trophic levels (Carpenter and Kitchell 1993) and humics constrain algal growth (Jones 1992). However, heavy phosphate loading, removal of macrophytes, overfishing, and removal of wetlands and riparian vegetation may lead to the pathological state in which there are few game fish, less grazing, no macrophytes, and extensive and frequent algal blooms (Harper 1992). This may be a rapid transition and it is not easily reversed (National Research Council 1992).
This situation appears to fit the definition of a hard loss of stability, because the change is rapid and large, and is sometimes not reversed even if phosphate loads are decreased (National Research Council 1992). One may say that the normal lake is resilient because it maintains its integrity under perturbation, but resilience is lost as phosphate loading and other stresses are increased. If critical levels of phosphate and other environmental variables could be identified, we might attempt to measure resilience in terms of the difference from the critical levels (Vollenweider 1976). Perhaps the question of whether or not the lake ecosystem fits our definitions may be answered by statistical analysis of long-term data.
The Baltic Sea Jansson and Velner (1995) describe the Baltic system in terms that show many similarities to the lake system. The Baltic Sea is partially enclosed and, consequently, has a residence time of water on the order of 20 years. Algae form the base of a diverse food web, with higher trophic levels occupied by commercially important species such as herring, flounder, pike, and perch. There may be long periods when there is weak vertical mixing of the water column, due to lack of inflow from the North Sea. During such periods, oxygen levels at greater depths may be very low and sulphur bacteria may predominate. The latter put large quantities of phosphorus into solution, which then upwell and cause plankton blooms.
In historical times, the Baltic has experienced several extended anoxic periods, but the system has not been permanently altered. Since the industrial revolution, the Baltic has been loaded with increasing amounts of phosphorus, and there are indications of a change of configuration to a detritus-based system. This would imply more turbid water and a fish community consisting mainly of sluggish species such as bream, roach, and ruffe, which are much less valuable than those previously listed. The possibility exists that the Baltic might reach a point at which even reducing phosphate inputs might not return the system to its earlier, more desirable state. Such a turn of events would correspond to a hard loss of stability, analogous to the behavior of the lake ecosystem. Unfortunately, there are no replicates of the Baltic system; hence, we have only analogies to guide action. A purposeful demonstration that the Baltic is actually capable of a sudden change corresponding to a hard loss of stability is unthinkable as an experiment. Nevertheless, it may be occurring as a result of human negligence. The earlier ability of the Baltic to recover from anoxic periods may correspond to resilience, but we cannot be sure whether this resilience is being lost.
The boreal forest Carpenter et al. (1996) characterize the boreal forest as a system with relatively few species and complicated interactions and dynamics. In upland regions, forests dominated by aspen and birch alternate with forests dominated by spruce and fir. Browsing by moose over a period of 20-40 years can convert an aspen stand into one dominated by conifers. As stands of conifers mature, they become increasingly favorable for reproduction of the spruce budworm. Eventually, outbreaks occur and portions of the system are converted into early successional aspen. The budworm outbreak corresponds to a hard loss of stability, and the combined upland system undergoes stable, long-period oscillations analogous to those described previously.
As the upland regions undergo these oscillations, the valley bottoms alternate between flooded plains and moist meadows. The flooded state is maintained by beavers, which cut aspen bordering streams for food and dam the streams to create ponds. When the supply of aspen is insufficient, the beavers abandon their dams, the dams break, and the ponds are soon replaced by meadows. This relatively rapid change, a consequence of decreasing supply of aspen, may be thought of as a hard loss of stability. The upland and lowland cycles tend to entrain each other because of the interaction between beavers and aspen. Fires also play a role in synchronizing cycles over large spatial areas, because conifers killed by the spruce budworm provide an abundance of fuel.
Although this system undergoes large alterations, sometimes very quickly, it may be thought of as resilient, maintaining its character over many centuries. Conditions at any given site may change abruptly, but the system is usually a mosaic of patches at differing stages of the cycle. When considered as a whole, it maintains considerable diversity.
Imagine starting with high levels of grass and low levels of woody vegetation. At low levels of stocking, there is only a small difference from the ungrazed system: if the system starts out with grass dominant, grass will continue to dominate. As stocking increases, the competition may favor woody vegetation. Eventually, there may be a collapse of the grass, and woody vegetation will dominate. Thus, the effect of grazing is to move the system from a state in which grass dominates to one in which woody vegetation dominates. Even when grazing pressure is relaxed, there may be little change in composition, because of the advantage enjoyed by woody vegetation over grass when the former is dominant. The effect of grazing is to move the system into the domain of attraction of woody vegetation for the ungrazed system.
If one plots grass density vs. the stocking level, the behavior may appear to be inexplicable: the grass level declines as grazing increases, but does not return to former levels when grazing returns to its former level. The apparent paradox is resolved if we realize that the density of grass depends not only on the stocking level, but also on competition with woody vegetation. These phenomena may be illustrated by a modification of the Lotka-Volterra competition model.
Mathematical model Let
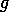 represent the
density of grass, and let
represent the
density of grass, and let 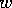 represent the density of woody
vegetation. The rate of change of grass density is assumed to be
represent the density of woody
vegetation. The rate of change of grass density is assumed to be
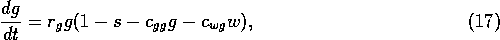
where
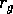 is a growth
rate, and
is a growth
rate, and 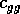 and
and
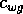 are competition
coefficients. The parameter
are competition
coefficients. The parameter  is determined by the stocking rate
of cattle. The rate of increase of the woody vegetation is assumed to
be
is determined by the stocking rate
of cattle. The rate of increase of the woody vegetation is assumed to
be
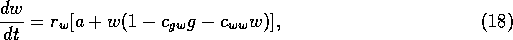
where
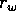 is a growth
rate,
is a growth
rate, 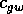 and
and 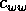 are competition
coefficients, and
are competition
coefficients, and  is a source term. In the illustrations that follow, the parameters
were chosen as:
is a source term. In the illustrations that follow, the parameters
were chosen as:
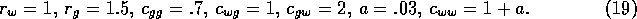
The case of light grazing corresponds to
 = 1/10.
= 1/10.
In order to understand the behavior of this system, it is helpful to
plot some curves in the 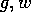 plane, as in Fig. 4
. In the phase
plane, the direction and speed of change of the system are given by
the vector
plane, as in Fig. 4
. In the phase
plane, the direction and speed of change of the system are given by
the vector  .
This vector is vertical on the curve where
.
This vector is vertical on the curve where 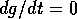 (the null
(the null 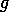 isocline), and it is horizontal on
the curve where
isocline), and it is horizontal on
the curve where 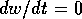 (the null
(the null 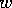 isocline). As can be seen,
isocline). As can be seen, 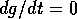 , where either
, where either 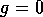 , or
, or

This locus is a straight line, and it shifts to the left as  increases. The null
increases. The null
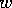 -isocline is a
hyperbola according to (Eq. 18). One of the asymptotes is the
-isocline is a
hyperbola according to (Eq. 18). One of the asymptotes is the 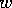 -axis, and the locus
passes through the point
-axis, and the locus
passes through the point 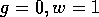 , labelled "W." This locus is
independent of the stocking parameter
, labelled "W." This locus is
independent of the stocking parameter  . Figure 4
shows in detail how a
system may approach more than one steady state, depending upon the
starting conditions. Although this system is much more complicated
than (Eq. 9), its qualitative behavior is the same if
. Figure 4
shows in detail how a
system may approach more than one steady state, depending upon the
starting conditions. Although this system is much more complicated
than (Eq. 9), its qualitative behavior is the same if  . This illustrates how
the very simple one-dimensional models may, nevertheless, be a
valuable heuristic guide.
. This illustrates how
the very simple one-dimensional models may, nevertheless, be a
valuable heuristic guide.
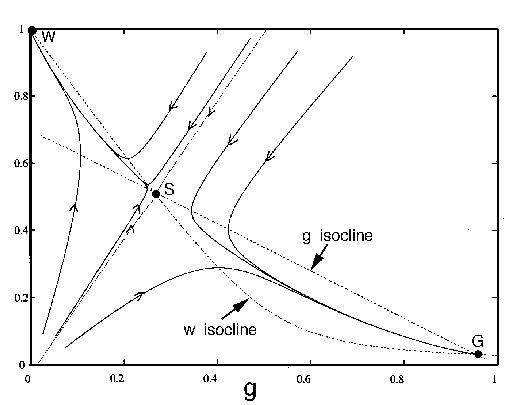
FIG. 4. The phase plane for grass and trees, according to
Eq. (17), Eq. (18), and Eq. (19). The null 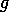 and
and 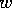 isoclines are plotted with dashed
lines. These are the curves where
isoclines are plotted with dashed
lines. These are the curves where 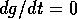 or
or 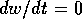 , respectively. The equilibria are
points where the isoclines cross. The stable equilibria are labelled
as "G'' and "W," respectively. The unstable equilibrium is labelled as
"S." The trajectories that enter S form a separatrix: they are
plotted with dotted lines. Trajectories to the right of the separatrix
eventually reach G, and those to the left eventually reach W.
, respectively. The equilibria are
points where the isoclines cross. The stable equilibria are labelled
as "G'' and "W," respectively. The unstable equilibrium is labelled as
"S." The trajectories that enter S form a separatrix: they are
plotted with dotted lines. Trajectories to the right of the separatrix
eventually reach G, and those to the left eventually reach W.
We now turn to the effect of increased stocking. The effect of an
increase in  is to
shift the null
is to
shift the null 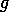 -isocline down and to the
left. Hence, the points S and G will approach each other along the
-isocline down and to the
left. Hence, the points S and G will approach each other along the
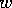 null
isocline. Because the separatrix passes through the point S, the
domain of attraction of G must shrink, whereas the domain of
attraction of W will expand. Qualitatively, the representation of
Fig. 4
still holds. For a still higher value,
null
isocline. Because the separatrix passes through the point S, the
domain of attraction of G must shrink, whereas the domain of
attraction of W will expand. Qualitatively, the representation of
Fig. 4
still holds. For a still higher value, 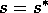 , the points S and G coincide. The
values of
, the points S and G coincide. The
values of 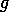 corresponding to the roots S and G are shown in Fig. 5
. A similar
diagram could be drawn to show the corresponding values of
corresponding to the roots S and G are shown in Fig. 5
. A similar
diagram could be drawn to show the corresponding values of 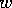 . This is a
bifurcation diagram analogous to Fig. 3
. If the stocking rate changes
slowly, we may expect the grass density to be given by the upper curve
in Fig. 5
. However, if
. This is a
bifurcation diagram analogous to Fig. 3
. If the stocking rate changes
slowly, we may expect the grass density to be given by the upper curve
in Fig. 5
. However, if 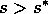 , the grass density must crash,
since there is no stable equilibrium with a nonzero grass
density. This is a hard loss of stability, and indeed there is a
striking similarity between Figs. 5
and 3.
, the grass density must crash,
since there is no stable equilibrium with a nonzero grass
density. This is a hard loss of stability, and indeed there is a
striking similarity between Figs. 5
and 3.
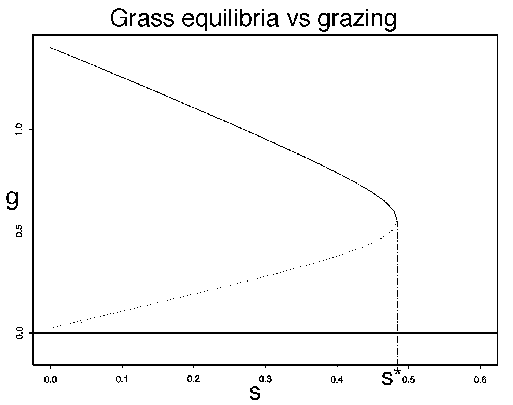
FIG. 5. A bifurcation diagram showing grass equilibria as a
function of the grazing parameter  for the system (17)-(19).If
for the system (17)-(19).If 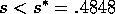 , there are three
roots. The lowest branch corresponds to the point W where
, there are three
roots. The lowest branch corresponds to the point W where 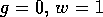 , which is always a
stable equilibrium. The middle branch corresponds to the locus of
point S as
, which is always a
stable equilibrium. The middle branch corresponds to the locus of
point S as  varies. Those points are unstable equilibria, so they are plotted with
a dotted line. The highest branch is the locus of point G as
varies. Those points are unstable equilibria, so they are plotted with
a dotted line. The highest branch is the locus of point G as  varies. This point is
always stable.
varies. This point is
always stable.
For values of 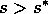 ,
the qualitative form of Fig. 6
applies. The only equilibrium is point
W, and all trajectories approach W. The domain of attraction of W is
the whole quadrant where
,
the qualitative form of Fig. 6
applies. The only equilibrium is point
W, and all trajectories approach W. The domain of attraction of W is
the whole quadrant where 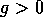 and
and 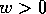 .
.
FIG. 6. The phase plane for grass and trees for the system
(17)-(19), with 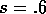 . In this case, there is only a
single stable equilibrium for the system at
. In this case, there is only a
single stable equilibrium for the system at 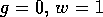 . All trajectories approach that
point.
. All trajectories approach that
point.

We may imagine the system beginning with the stocking rate 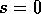 . According to Fig. 5
,
the unstable equilibrium S and the stable equilibrium W are very close
together. Hence, the separatrix in a figure analogous to Fig. 4
is in
the extreme upper left corner: virtually any initial combination of
. According to Fig. 5
,
the unstable equilibrium S and the stable equilibrium W are very close
together. Hence, the separatrix in a figure analogous to Fig. 4
is in
the extreme upper left corner: virtually any initial combination of
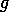 and
and 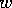 will lead to a high
density of
will lead to a high
density of 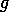 , given
by the upper branch in Fig. 5
, with a correspondingly small density of
, given
by the upper branch in Fig. 5
, with a correspondingly small density of
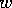 . Now, if
. Now, if  is increased slowly,
the density of
is increased slowly,
the density of 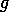 will move downward along the upper branch until the bifurcation point
will move downward along the upper branch until the bifurcation point
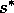 is
reached. Beyond that point, the grass density must crash, and woody
plants will dominate. Even if grazing pressure is withdrawn (
is
reached. Beyond that point, the grass density must crash, and woody
plants will dominate. Even if grazing pressure is withdrawn (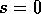 ), the grass cannot
recover because it will be to the left of the separatrix. This is the
hysteresis effect.
), the grass cannot
recover because it will be to the left of the separatrix. This is the
hysteresis effect.
When viewed at a long time scale, a brief period of grazing may cause a rapid collapse of the grass, followed by a slow recovery. What appears to be an equilibrium with high woody vegetation, when viewed at a short time scale, corresponds to a region in which the system spends a long time when viewed at a long time scale. This sort of qualitative behavior is analogous to "excitable systems." Such systems are best known as models of the nerve impulse, according to the theory of Hodgkin and Huxley, as modified by Fitzhugh. The system has a single stable equilibrium, but when perturbed in an appropriate direction, it may undergo a very large excursion (firing of the neuron), followed by a long recovery (refractory) period. Details are given in Edelstein-Keshet (1988).
The effect of fire on dynamics To model aging properly is complicated, but for present purposes, it suffices to find a simple system that has the required qualitative behavior. We do not contend that the following model is an accurate representation of the true dynamics. We must keep track of surplus grass that may serve as fuel for fires. Hence, we let gross grass production be given by
 , where
, where

where
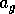 is a source
term, and
is a source
term, and 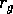 is the
rate of grass production available for grazing. The grass that is not
consumed by grazing cattle is potential fuel for fires, and is denoted
by
is the
rate of grass production available for grazing. The grass that is not
consumed by grazing cattle is potential fuel for fires, and is denoted
by 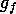 , where
, where

The parameter
 determines the proportion of grass consumed by cattle. Now (Eq.17) is
replaced by
determines the proportion of grass consumed by cattle. Now (Eq.17) is
replaced by
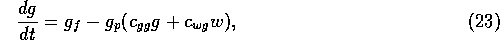
The dynamics of
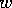 will be influenced by fires, and the age of trees influences their
susceptibility to fire. Let a new variable
will be influenced by fires, and the age of trees influences their
susceptibility to fire. Let a new variable  denote the product of the woody
plant density and the average age of the woody plants. A first
approximation yields
denote the product of the woody
plant density and the average age of the woody plants. A first
approximation yields 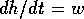 , but that relationship neglects
the influence of fire on the average age. The dynamics of
, but that relationship neglects
the influence of fire on the average age. The dynamics of 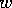 and
and  are given by
are given by
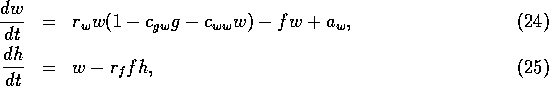
where the fire risk
 is defined as follows: let the fire
potential
is defined as follows: let the fire
potential 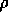 be
proportional to the available fuel:
be
proportional to the available fuel:
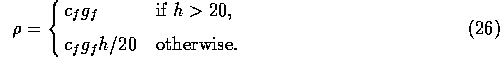
We assume that the fire risk
 is given by
is given by

where the parameter
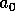 is an age at which the fire risk is
half of its maximum, and the parameter
is an age at which the fire risk is
half of its maximum, and the parameter 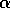 determines the sharpness of the
increase of fire risk with age. We have used
determines the sharpness of the
increase of fire risk with age. We have used 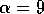 to give a sharp increase, and
to give a sharp increase, and 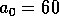 . The remaining
parameters were chosen as
. The remaining
parameters were chosen as
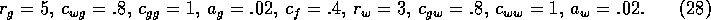
The stocking rate
 may be chosen as a parameter or control variable. In the following, we
chose
may be chosen as a parameter or control variable. In the following, we
chose 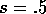 .
.
Because there are three state variables in this system, one cannot
make a meaningful phase plot. However, if the age variable  is fixed, we may gain
an impression of the dynamics. Figure 7
shows how a low value of
is fixed, we may gain
an impression of the dynamics. Figure 7
shows how a low value of  leads the system to an
equilibrium with high
leads the system to an
equilibrium with high 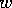 . On the other hand, a higher
value of
. On the other hand, a higher
value of  leads to
an equilibrium with much lower
leads to
an equilibrium with much lower 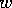 , as shown in Fig. 8
. Now, if
, as shown in Fig. 8
. Now, if  increases with time,
the system will first have high
increases with time,
the system will first have high 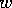 , then lower
, then lower 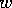 as it ages. Fires decrease the
average age of trees as well as their density. Hence, the system gets
reset to a state analogous to that shown in Fig. 7
after a fire. The
cycling of
as it ages. Fires decrease the
average age of trees as well as their density. Hence, the system gets
reset to a state analogous to that shown in Fig. 7
after a fire. The
cycling of 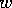 with
time in the full system, with
with
time in the full system, with  changing, is shown in Fig. 9
.
changing, is shown in Fig. 9
.
FIG. 7. The phase plane for grass and trees for the system
(21)-(28), neglecting the dynamics of the age variable 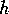 , with a low value of
, with a low value of
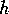 . There is only a
single equilibrium with a high value of
. There is only a
single equilibrium with a high value of 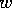 and a lower value for
and a lower value for 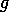 , and it is stable. All
trajectories approach this equilibrium.
, and it is stable. All
trajectories approach this equilibrium.
FIG. 8. The phase plane for grass and trees for the system
(21)-(28), neglecting the dynamics of the age variable 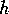 , with a high value of
, with a high value of
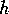 . The single
equilibrium has a low value for
. The single
equilibrium has a low value for 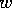 and a moderately high value for
and a moderately high value for 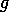 .
.
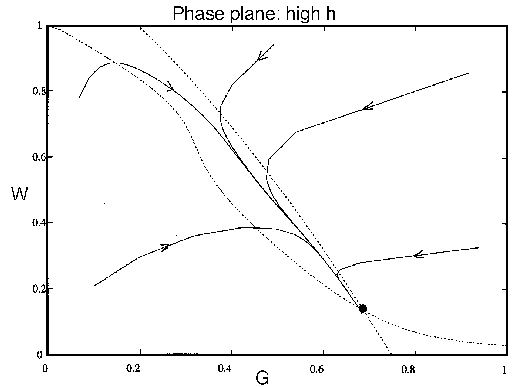
FIG. 9. Tree density (W) vs. time (T, in
years) for the system (21)-(28), including dynamics for the age
variable. A similar figure could be drawn for the grass density, which
tends to be high when woody vegetation is low, and vice versa.
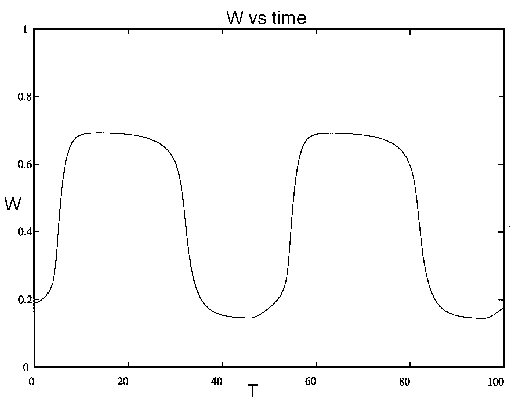
If one were to observe this system over a time of 5-20 years, it would
appear that woody plants would eventually dominate grasses because of
the combination of competition and grazing. Over the next 30 years,
however, the effect of fire and aging of trees leads to a collapse of
the trees, making way for the next cycle. This example illustrates how
the time scale over which we observe the system may have a decisive
influence upon our classification of its behavior. Unfortunately, the
scale over which we are able to observe the system is often much
shorter than the scale over which it exhibits its characteristic
behavior.
Here, we have proceeded from simple conceptual models, such as the
raft analogy, to simple but abstract mathematical models, to ecosystem
analogues, and finally to a fairly detailed model of a savanna
system. In no case can these analogies be considered to be complete,
nor is our knowledge of the systems detailed enough to support a
full-blown model with statistical justification. Perhaps one might
conclude that all this is merely speculation, unworthy of serious
attention. On the other hand, prudent decision making requires that we
take account of a variety of plausible hypotheses about the responses
to our actions. The examples presented here do not encourage
complacency about the ability of natural systems to support us and our
habits in the lavish fashion we have enjoyed in the past. If we refuse
to contemplate possibilities that are only dimly perceived, we may
miss the opportunity to learn about the world and adapt our behavior
accordingly. If we insist that the simplest and most convenient
hypotheses have priority when choosing actions, we run the risk of
allowing sloth and pride to bring us to disaster.
Responses to this article are invited. If accepted for publication, your response will be hyperlinked to the article. To submit a comment, follow this link. To read comments already accepted, follow this link.
This research was the product of a Resilience Network Planning Workshop supported by the Beijer Institute, a Division of the Royal Swedish Academy of Sciences.
- Arrow, K., B. Bolin, R. Costanza, P. Dasgupta, C. Folke, C. S. Holling, B.-O. Janssen, S. Levin, K.-. Mäler, C. Perrings, and D. Pimentel. 1995. Economic growth, carrying capacity, and the environment. Science 268: 520-521.
- Carpenter, S. R., B. O. Jannson, D. Ludwig, J. Pastor, G. Peterson, and B. Walker. 1996. A comparative analysis of total system resilience. Notes from the Beijer Institute Resilience Network Planning Workshop April 10-16.
- Carpenter, S. R., and J. F. Kitchell. 1993. The trophic cascade in lakes. Cambridge University Press, Cambridge, UK.
- Carpenter, S. R., and P. R. Leavitt. 1991. Temporal variation in the paleolimnological record arising from a trophic cascade. Ecology 72(1): 277-285.
- D'Antonio, C. M., and P. M. Vitousek. 1992. Biological invasions by exotic grasses, the grass-fire cycle, and global change. Annual Review of Ecology and Systematics 23: 63-87.
- Done, T. J. 1992. Phase shifts in coral reef communities and their ecological significance. Hydrobiologia 247: 121-132.
- Dublin, H. T., A. R. E. Sinclair, and J. McGlade. 1990. Elephants and fire as causes of multiple stable states in the Serengeti-Mara woodlands. Journal of Animal Ecology 59: 1147-1164.
- Edelstein-Keshet, L. 1988. Mathematical models in biology. Random House, New York, New York, USA.
- Estes, J. A., and D. Duggins. 1995. Sea otters and kelp forests in Alaska: generality and variation in a community ecological paradigm. Ecological Monographs 65(1): 75-100.
- Guckenheimer, J., and P. Holmes. 1983. Nonlinear oscillations, dynamical systems, and bifurcations of vector fields. Springer-Verlag, New York, New York, USA.
- Harper, D. 1992. Eutrophication of freshwaters. Chapman and Hall, London, UK.
- Holling, C. S. 1973. Resilience and stability of ecological systems. Annual Review of Ecology and Systematics 4: 1-23.
- Hughes, T. P. 1994. Catastrophes, phase shifts, and large-scale degradation of a Caribbean coral reef. Science 265: 1547-1551.
- Jansson, B.-O., and H. Velner. 1995. The Baltic: the sea of surprises. Pages 292-372 in L. H. Gunderson, C. S. Holling, and S. S. Light, editors. Barriers and bridges to the renewal of ecosystems and institutions. Columbia University Press, New York, New York, USA.
- Jones, R. I. 1992. The influence of humic substances on lacustrine planktonic food chains. Hydrobiologia 229: 73-91.
- Ludwig, D., D. Jones, and C. S. Holling. 1978. Qualitative analysis of insect outbreak systems: the spruce budworm and the forest. Journal of Animal Ecology 47: 315-332.
- McClanahan, T. R., A. T. Kamukuru, N. A. Muthiga, M. Gilagabher Yebio, and D. Obura. 1996. Effect of sea urchin reductions on algae, coral, and fish populations. Conservation Biology 10(1): 136-154.
- National Research Council. 1992. Restoration of aquatic ecosystems. National Academy Press, Washington, D.C., USA.
- Odum, E. P. 1993. Ecology and our endangered life support systems. Second edition. Sinauer, Sunderland, Massachusetts, USA.
- Pimm, S. L. 1991. The balance of nature? University of Chicago Press, Chicago, Illinois, USA.
- Scheffer, M., S. H. Hosper, M.-L. Meijer, B. Moss, and E. Jeppesen. 1993. Alternative equilibria in shallow lakes. TREE 8(8): 275-279.
- Schindler, D. W. 1990. Experimental perturbations of whole lakes as tests of hypotheses concerning ecosystem structure and function. Oikos 57: 25-41.
- Vollenwieder, R. A. 1976. Advances in defining critical loading levels for phosphorus in lake eutrophication. Memorie dell'Istituto Italiano di Idrobiologia 33: 53-83.
- Walker, B. H., D. Ludwig, C. S. Holling, and R. M. Peterman. 1981. Stability of semiarid savanna grazing systems. Journal of Ecology 69:473-498.
- Zimov, S. A., V. I. Chuprynin, A. P. Oreshko, F. S. Chapin, III, J. F. Reynolds, and M. C. Chapin.1995. Steppe-tundra transition: a herbivore-driven biome shift at the end of the Pleistocene. American Naturalist 146(5): 765-794.
- Carpenter, S. R., B. O. Jannson, D. Ludwig, J. Pastor, G. Peterson, and B. Walker. 1996. A comparative analysis of total system resilience. Notes from the Beijer Institute Resilience Network Planning Workshop April 10-16.
Return times and resilience It is important to distinguish between behavior near a stable equilibrium and behavior near the boundary of a domain of attraction, which is an unstable equilibrium or separatrix. As discussed in Section 2, the long return times associated with a loss of resilience are caused by slow dynamics near the unstable equilibrium, not by slow dynamics near the stable equilibrium point. Unfortunately, there are two conflicting definitions of resilience and consequent confusion about the connection between resilience and return times.
Pimm (1991:13) defines resilience as "how fast a variable that has
been displaced from equilibrium returns to it. Resilience could be
estimated by a return time, the amount of time taken for the
displacement to decay to some specified fraction of its initial
value.'' Pimm (1991: 33) describes return to equilibrium by the
equation

where 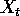 is the
population density at time
is the
population density at time 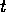 ,
, 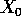 is the initial population density,
and
is the initial population density,
and 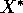 is the
equilibrium density. The differential equation for
is the
equilibrium density. The differential equation for 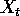 that corresponds to this formula is
that corresponds to this formula is

A similar model with discrete time could be given instead, but that
would not alter the following argument. If we measure displacement
from 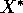 by
by  , then
, then  satisfies
satisfies

which is equivalent to Eq. (A.1) if 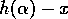 is replaced by
is replaced by  . Strictly speaking,
Pimm's definition depends upon this simplicity, because the amount of
time required for
. Strictly speaking,
Pimm's definition depends upon this simplicity, because the amount of
time required for  to decay to some specified fraction of its initial value is only
constant if the model (A.1) is used. In
fact, if the initial displacement is
to decay to some specified fraction of its initial value is only
constant if the model (A.1) is used. In
fact, if the initial displacement is 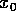 and the fraction is
and the fraction is 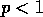 , then (A.1) implies that
, then (A.1) implies that

from which we conclude that the return time 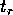 is given by
is given by

The remarkable feature is that the magnitude 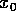 does not appear in this
formula. This is a feature of this model only, as we shall see
below. In more general circumstances, such a result can be expected to
hold only in the limit as
does not appear in this
formula. This is a feature of this model only, as we shall see
below. In more general circumstances, such a result can be expected to
hold only in the limit as 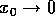 . Such results are called "local."
As pointed out in Section 2, a common error is to extrapolate local
results to global ones. In the present context, it amounts to
replacing a complicated function by a linear approximation. Such
approximations are certainly easy to work with, but they may miss
essential features of the dynamics. In fact, failure to recognize the
distinction between local stability and global stability can lead to
unwarrented optimism about the likely consequences of interventions in
natural systems. If we think that stability to small perturbations
necessarily implies stability to large perturbations, then precautions
are never required.
. Such results are called "local."
As pointed out in Section 2, a common error is to extrapolate local
results to global ones. In the present context, it amounts to
replacing a complicated function by a linear approximation. Such
approximations are certainly easy to work with, but they may miss
essential features of the dynamics. In fact, failure to recognize the
distinction between local stability and global stability can lead to
unwarrented optimism about the likely consequences of interventions in
natural systems. If we think that stability to small perturbations
necessarily implies stability to large perturbations, then precautions
are never required.
In order to distinguish behavior near the equilibrium at 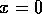 from behavior near an
unstable equilibrium, we must use a model with more parameters than (A.2). We set
from behavior near an
unstable equilibrium, we must use a model with more parameters than (A.2). We set
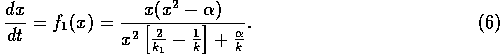
This particular form leads to an especially simple equation for the
return time: the time to reach a position 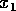 starting at
starting at 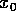 is given by
is given by

and the form for 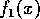 was chosen so that
was chosen so that
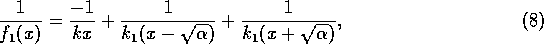
as can be verified algebraically. In view of (A.7) and (A.8),
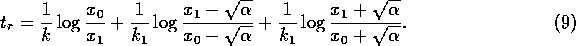
Now, if we replace 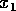 by
by 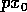 , (A.9)
becomes
, (A.9)
becomes
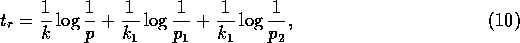
where

If the last two terms in (A.10) are
omitted, this result is identical to Pimm's assumption (A.1). Our more complicated dynamical
assumption (A.6) is the analogue of Pimm's
assumption if there are three equilibria. Under what conditions does
(A.10) imply large return times? The first
term, which corresponds to Pimm's model, implies a long return time if
the ratio 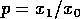 is
small or if
is
small or if 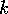 is
small. In Pimm's discussion,
is
small. In Pimm's discussion, 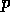 is a parameter that describes a
probe or observation of the system. Ordinarily,
is a parameter that describes a
probe or observation of the system. Ordinarily, 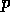 is fixed, and the return time
provides an estimate for
is fixed, and the return time
provides an estimate for 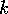 .
.
The second term in (A.10) implies a long
return time if 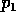 is
small or
is
small or  is
small. Our previous discussion was concerned with a possibly variable
is
small. Our previous discussion was concerned with a possibly variable
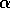 and disturbances
that might take the system near an unstable equilibrium. That
corresponds to
and disturbances
that might take the system near an unstable equilibrium. That
corresponds to 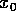 near
near 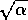 , or
, or 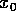 near
near 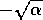 . In such a case,
. In such a case, 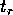 will be large even if the parameter
will be large even if the parameter
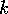 is large. That
is, return times may be long, even for systems that show very rapid
return when close to the stable equilibrium. According to this point
of view, long return times may be diagnostic for a small
is large. That
is, return times may be long, even for systems that show very rapid
return when close to the stable equilibrium. According to this point
of view, long return times may be diagnostic for a small 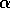 or for disturbances
that are large enough to take the system near an unstable
equilibrium. They may also correspond to weak repulsion from the
unstable equilibrium, i. e., small
or for disturbances
that are large enough to take the system near an unstable
equilibrium. They may also correspond to weak repulsion from the
unstable equilibrium, i. e., small  . If a disturbance takes the
system beyond the unstable equilibrium, there is no return at all.
. If a disturbance takes the
system beyond the unstable equilibrium, there is no return at all.
In summary, according to Pimm (1991) and according to us, long return
times may be diagnostic for a loss of resilience, but the meanings of
the terms are quite different in the two cases. Pimm is concerned with
behavior near a stable equilibrium. In that case, a long return time
for a given displacement from the equilibrium indicates a small
coefficient 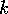 or,
equivalently, a small derivative of
or,
equivalently, a small derivative of 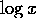 . We are concerned with behavior
of a system with two or three equilibria, one of which is
stable. Resilience describes the tendency of the system to return to
its stable equilibrium. A long return time is due to disturbances that
bring the system near an unstable equilibrium, or possibly to a weak
repulsion from an unstable equilibrium.
. We are concerned with behavior
of a system with two or three equilibria, one of which is
stable. Resilience describes the tendency of the system to return to
its stable equilibrium. A long return time is due to disturbances that
bring the system near an unstable equilibrium, or possibly to a weak
repulsion from an unstable equilibrium.
Address of Correspondent:
Donald Ludwig
Department of Mathematics
University of British Columbia
Vancouver, British Columbia, Canada V6T 1Z2
phone: 604-541-9409
fax: 604-822-6074
ludwig@math.ubc.ca
Addresses of Coauthors:
Brian Walker
CSIRO, Division of Wildlife and Ecology
G.P.O. Box 84
Lynham, ACT 2602, Australia
Phone: 6-242-1742
Fax: 6-241-1742
brian.walker@dwe.csiro.au
Crawford S. Holling
University of Florida
Department of Zoology, P.O. Box 118525
Gainesville, Florida 32611 USA
Phone: 352-392-6914
Fax: 352-392-3704
holling@zoo.ufl.edu
*The copyright to this article passed from the Ecological Society of America to the Resilience Alliance on 1 January 2000.