|
|
|
Copyright © 2000 by The Resilience Alliance
The following is the established format for referencing this article:
Dawe, N. K., G. E. Bradfield, W. S. Boyd, D. E. C. Trethewey, and A. N. Zolbrod. 2000. Marsh creation in a northern Pacific estuary: Is thirteen years of monitoring vegetation dynamics enough? Conservation Ecology 4(2): 12. [online] URL: http://www.consecol.org/vol4/iss2/art12/
A version of this article in which text, figures, tables, and appendices are separate files may be found by following this link.
Report Marsh Creation in a Northern Pacific Estuary: Is Thirteen Years of Monitoring Vegetation Dynamics Enough? Neil K. Dawe1, Gary E. Bradfield2, W. Sean Boyd1, Donald E. C. Trethewey1, and A. Nana Zolbrod2
1Canadian Wildlife Service; 2University of British Columbia
- Abstract
- Introduction
- Methods
- Results
- Discussion
- Conclusions
- Responses to this Article
- Acknowledgments
- Literature Cited
- Appendix 1
Vegetation changes were monitored over a 13-yr period (1982-1994) in the Campbell River estuary following the development of marshes on four intertidal islands. The marshes were created to mitigate the loss of a natural estuarine marsh resulting from the construction of a dry land log-sorting facility. Plant species coverage was measured along 23 permanent transects in planted and unplanted blocks on the constructed islands, and in naturally occurring low-marsh and mid-to-high marsh reference communities on nearby Nunn’s Island. Five dominant species, Carex lyngbyei, Juncus balticus, Potentilla pacifica, Deschampsia caespitosa, and Eleocharis palustris established successfully and increased in cover in both planted and unplanted areas. The planted, unplanted, and Nunn’s Island low-marsh sites had similar total plant cover and species richness by the 13th year. Principal components analysis of the transects through time indicated successful establishment of mid-to-low marsh communities on the constructed islands by the fourth year. Vegetation fluctuations on the constructed islands were greater than in the mid-to-high and low-marsh reference communities on Nunn’s Island. Results showed that substrate elevation and island configuration were major influences on the successful establishment and subsequent dynamics of created marsh communities. Aboveground biomass estimates of marshes on the created islands attained those of the reference marshes on Nunn’s Island between years 6 and 13. However, Carex lyngbyei biomass on the created islands had not reached that of the reference marshes by year 13. Despite the establishment of what appeared to be a productive marsh, with species composition and cover similar to those of the reference marshes on Nunn’s Island, vegetation on the created islands was still undergoing changes that, in some cases, were cause for concern. On three of the islands, large areas devoid of vegetation formed between years 6 and 13, probably a result of water ponding. Adaptive management has allowed us to modify the island configuration through the creation of channels to drain these sites in an attempt to reverse the vegetation dieback. These changes, occurring even after 13 years, further underscore the need for caution when considering the trading of existing natural, healthy, productive wetlands for the promise of created marshes that may or may not prove to be equal to the natural systems. Where marsh creation is warranted, we recommend that management of created marshes be adaptive and flexible, including a long-term monitoring program that should continue at least until the annual variation in vegetation of the created marsh is similar to that of natural, nearby systems.
KEY WORDS: adaptive management of ecosystems, Carex lyngbyei, Deschampsia caespitosa, Eleocharis palustris, estuarine marsh creation, Juncus balticus, long-term vegetation dynamics, mitigation cautions, natural vs. constructed wetlands, Potentilla pacifica, restoration ecology, wetland creation.
Published: December 15, 2000
Restoration ecology involves the development of structural or functional characteristics of ecosystems that have been lost (Cairns 1988). It includes habitat creation that aims to establish plant communities that are representative of the original, undamaged state (Buckley 1989). Many restoration projects are implemented as mitigation for the loss of natural marshes resulting from development. In some cases, resource agencies have adopted a “no net loss” policy that requires the proponent to create replacement marsh habitat in some ratio of created to lost habitat, such as 2:1. Although this may seem reasonable, it assumes that enough is known about natural wetland systems that they can be created with all of their attendant functions. Because of the belief that we can build wetlands based on past, apparently successful attempts, it has also meant that the resource agencies often approve development proposals that destroy diverse, productive wetlands, rather than simply protecting the integrity of these natural wetland systems. Unfortunately, the apparently successful attempts have often been monitored for only a short time period (usually 1-2 years), if at all.
Coastal marsh restoration on the east coast of the United States has occurred to a far greater extent than on the Pacific coast, with mixed success (Seneca et al. 1976, Broom et al. 1986, Mason and Slocum 1987). On the U.S. Pacific coast, marsh restoration and creation projects have been carried out from San Diego to the Pacific Northwest (e.g., Zedler 1984, Frenkel and Morlan 1991, Zedler and Langis 1991, Simensted and Thom 1996). In British Columbia, many coastal marshes are in need of restoration because they have suffered heavy damage from past human activities, and are critical habitats for sustaining healthy populations of fish, migratory birds, and other wildlife (Butler et al. 1989, Levings and Macdonald 1991). Recently, many restoration projects have been undertaken in the estuaries of southwestern British Columbia. They have involved both restoration of degraded marshes and creation of new marsh habitat, principally as mitigation for development (Anonymous 1995).
On Vancouver Island, studies of vegetation change following the breaching of sea dikes to restore natural tidal flushing have been reported by Dawe and Jones (1986), Campbell and Bradfield (1988), and Dawe and McIntosh (1993). On the adjacent mainland, in the Fraser River estuary, several projects aimed at increasing the area of intertidal marshland have been undertaken since the early 1980’s (Kistritz 1995, Adams and Williams, in press). Varying degrees of success were achieved, but even in the “successful” cases, species diversity and total vegetation cover remain below those of comparable, naturally occurring marshes. Improper design features relating to surface elevation and exposure to strong currents, as well as intense grazing by Canada Geese, were the major reasons that the created marshes failed.
Because our knowledge of these complex systems is poor (cf., Holling and Meffe 1996, Lee 1999), the adoption of a conservative management approach is warranted. Holling and Meffe (1996) suggest that the default condition of ecosystem management, unless proven otherwise, should be the retention of the natural state rather than any manipulation of system components or dynamics. When manipulation is deemed necessary, adaptive management should be encouraged. Adaptive management is a structured, continual process of “learning by doing,” (Holling 1978, Walters 1986, Christensen et al. 1996). Management objectives are clearly established based on the best available science, and the results are monitored to provide timely feedback to managers, who may then modify their activities accordingly. As Hilborn (1992) notes, “If decisions are not made based on the results of monitoring and evaluation, learning will not take place.”
Monitoring over the long term is an important component of adaptive management. Time scales for system components to respond to management prescriptions are typically long (Lee 1999) because they often deal with slowly changing variables. Also, systems are continuously changing through natural processes, making it difficult to determine their “normal state.” As a result, our knowledge of them is always incomplete (Walters and Holling 1990, Ehrenfeld 1991). In addition to the natural variation in these systems, there are the added changes they undergo as the effects of human activities, which include the management activities, interact with them. It then becomes difficult to distinguish the effects of management from those of natural variation in the system (Walters and Holling 1990). Thus, ecosystems are “moving targets,” having uncertain and unpredictable futures (Holling 1978, Walters 1986).
One of the largest marsh creation projects undertaken in British Columbia occurred in the Campbell River estuary in 1981 (Brownlee et al. 1984). The project involved the construction of four intertidal islands with a total plantaed surface area of 2.4 ha. Over 23,000 marsh vegetation plugs salvaged from the construction of a nearby log-sorting facility and holding pond were transplanted to the islands. An important adaptive component of the project was a long-term monitoring study of the vegetation growth on the islands. Although small-scale projects such as this have not received as much attention in the literature as have large-scale issues (e.g., management of a fishery), Johnson (1999) notes that there are many applications for an adaptive management approach, including wetland restoration.
Our paper summarizes the results of a monitoring study of the developing marsh communities in the Campbell River estuary conducted over a 13-yr period. The objectives were: (1) to evaluate the success of the marsh creation project when compared with a nearby, natural system and to be able to react to unforseen problems that may arise; and (2) to determine the degree to which the vegetation changes through time were related to substrate elevation, planting design, and island configuration. Long-term comparative studies such as this have rarely been reported for intertidal marshes along the northern Pacific coast (Race 1985); the results will be useful for other rehabilitation efforts in these ecosystems.
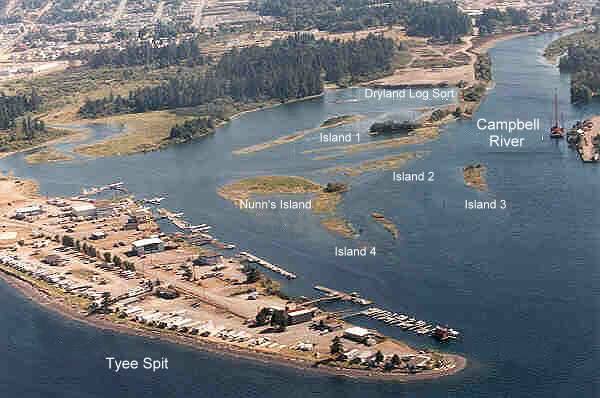
The Campbell River estuary is located on the central eastern coast of Vancouver Island (50° 02’ N, 125° 15’ W; Figs. 1 and 2). Much of the estuarine habitat is situated behind Tyee Spit, which supports an area of light industry (Fig. 1). The estuary also has two marinas plus an active float plane base. From a watershed covering 1461 km2, the river has the third largest discharge on the east coast of Vancouver Island (mean: 108 m3 /s). The river flows have been regulated since 1949 by the John Hart dam.
|
Fig. 1. Aerial photo of the Campbell River estuary looking south. Shown are the constructed islands in relation to Tyee Spit, Nunn’s Island, and the dry land log-sort area (6 August 1998).
|
The mean and greatest tidal ranges in the estuary are 2.9 m and 4.6 m, respectively; there are two complete tidal oscillations daily (Bell and Thompson 1977). Temperature and precipitation data for the Campbell River area show that the first part of the growing season (April - June) tends to be cooler and wetter (mean monthly temperature 14.7oC; mean monthly precipitation 51.4 mm) than the latter part of the growing season (15.5oC; 39.8 mm; July-September). Dormant season months (October-March) tend to be cool and wet (6.2oC, 160.3 mm; Anonymous 1993).
Between 1904 and 1981, the estuary was used primarily as a log storage and sorting area. As a result, the benthos became completely covered with woody debris and waste and, thus, was no longer biologically productive habitat. In 1981, the intertidal area used for storage and sorting of logs was reduced from 32.8 ha to 6.8 ha (Brownlee et al. 1984) because of the construction of a dry land log-sorting facility.
The Campbell River estuary provides important habitat for five species of Pacific salmon (Onchorhynchus spp.), steelhead (O. mykiss) and cutthroat (O. clarki) trout, and Dolly Varden char (Salvelinus malma; Brownlee et al. 1984), and supports both migrant and wintering populations of migratory birds (Dawe et al. 1995).
Experimental design and planting treatmentsThe marsh construction project was undertaken in 1981 as part of an agreement requiring British Columbia Forest Products Ltd. (now TimberWest Forest Ltd.) to compensate for the loss of about 0.42 ha of intertidal marshes through the expansion of their log-sorting facilities. Scientific aspects of the project, including island design and transplanting methods, were overseen by biologists from Fisheries and Oceans Canada and the Canadian Wildlife Service of Environment Canada. Four islands were constructed in the winter of 1981-1982 from dredge spoils recovered from the creation of the dry land log-sort holding pond. The islands produced a total surface area (excluding side slopes) of about 2.4 ha (Fig. 2). The islands were designed to optimize planting success, taking into consideration elevation, slope, and substrate stability (Brownlee et al. 1984). Substrate elevations of islands 1, 2, and 4 were graded to approximately 3.0 m above chart datum (ACD), considered to be optimum for growth of Carex lyngbyei in the estuary. C. lyngbyei is a productive sedge with high habitat values for juvenile fish and migratory birds. Island 3 was graded from subtidal to about 4.0 m ACD, which was similar to the elevational gradient on Nunn’s Island, the naturally occurring reference island in the estuary.
Over 23,000 marsh “plugs” containing substrate, plant rhizomes and roots, and aerial shoots were placed into planting blocks (size range: 300 m2 to 1,100 m2) on the constructed islands during the nocturnal, low tides of February 1982. The plugs measured approximately 15 x 15 x 20 cm deep. They were cut from larger mats collected during the previous November from a donor site in the southeastern part of the estuary, and were moved to an overwintering site near island 1. The plugs were planted at two randomly selected densities in the planting blocks on each island: 0.5-m or 1.0-m intervals (Table 1). Some areas on the islands were left unplanted to assess the extent and rate of natural colonization. In total, about 0.8 ha were planted on the four constructed islands (Brownlee et al. 1984).
|
Table 1. Mean elevation (ACD is above chart datum), treatments, and inundating water salinities of permanent transects on marsh transplant islands in the Campbell River estuary, British Columbia.
|
Unfortunately, neither species cover nor elevations at the donor site (which was destroyed in creating the log-holding pond) were recorded before the vegetation mats were cut and moved to the overwintering site. However, the dominant species noted at the donor marsh included Carex lyngbyei, Juncus balticus, Equisetum spp., Potentilla pacifica, and Deschampsia caespitosa (Brownlee et al. 1984). With the exception of Equisetum spp., these species are typical of low-salinity, estuarine marshes in south-coastal British Columbia. The species composition of the planted material suggests that the donor site elevation was in the mid-marsh range (Dawe and White 1982).
Sampling
In late June 1982, 23 permanent transects (30-80 m in length) were established, running diagonally through the transplanted and unplanted blocks (Fig. 2 and
Sampling surveys of all transects were carried out during late June or early July of 1982, 1983, 1984, 1985, 1986, 1988, and 1994. For the first two years, the success of individual transplant plugs was monitored; by 1984, it became too difficult to distinguish the individual plugs over much of the study area. Plugs were considered successful if they contained at least one living vascular plant.
Vegetation data were collected by systematically sampling 1.0 m2 relevés
spaced at 5.0-m intervals along the permanent transects as described in Dawe and White (1982, 1986). Cover classes of vascular plant species were recorded using the Braun-Blanquet cover-abundance scale (Mueller-Dombois and Ellenberg 1974). Although the Braun-Blanquet scale tends to minimize differences between observers in cover estimates, such differences may arise, particularly when the actual cover is close to the class boundaries (i.e., 25%, 50%, 75%). In this study, the senior author participated in all sampling surveys to ensure consistency. Nomenclature follows Hitchcock and Cronquist (1973).
Transects 10 and 11 on island 3 were not sampled in 1994, as they were considerably higher in elevation than the other transects, and were developing into upland communities, not representative of intertidal marsh vegetation.
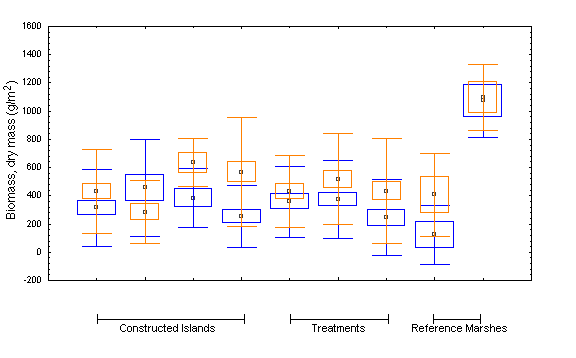
In 1988 and 1994, estimates of aboveground biomass were obtained. Five 0.125-m2 vegetation samples were randomly taken from each of the planted and
unplanted blocks and the Nunn’s Island reference marshes. All living vegetation was clipped with garden shears at substrate level within the sample frame, and was placed in opaque plastic bags. The collected samples were stored in a freezer until they could be analyzed.
In the laboratory, the individual samples were processed as outlined in Eilers (1975). Briefly, they were separated into their constituent species fractions, the number of stems of each species was determined, and the species fraction was weighed to the nearest 0.1 g on a triple beam balance. The species fraction was then cut into small pieces and placed in an open paper bag in a ventilated drying oven set at 85°C. Subsamples were taken where the species fraction
exceeded about 50 g. The samples and subsamples were dried until constant mass was attained; subsample mass values were converted to total dry mass by using a simple proportion calculation.
On the high tide of 26 June 1984, the salinity of the inundating waters over each block was determined using a conductivity meter. Measurements were gathered to determine if salinity of the inundating waters could be a factor in the poor growth of the vegetation at the lower elevations of island 3.
Data analysis
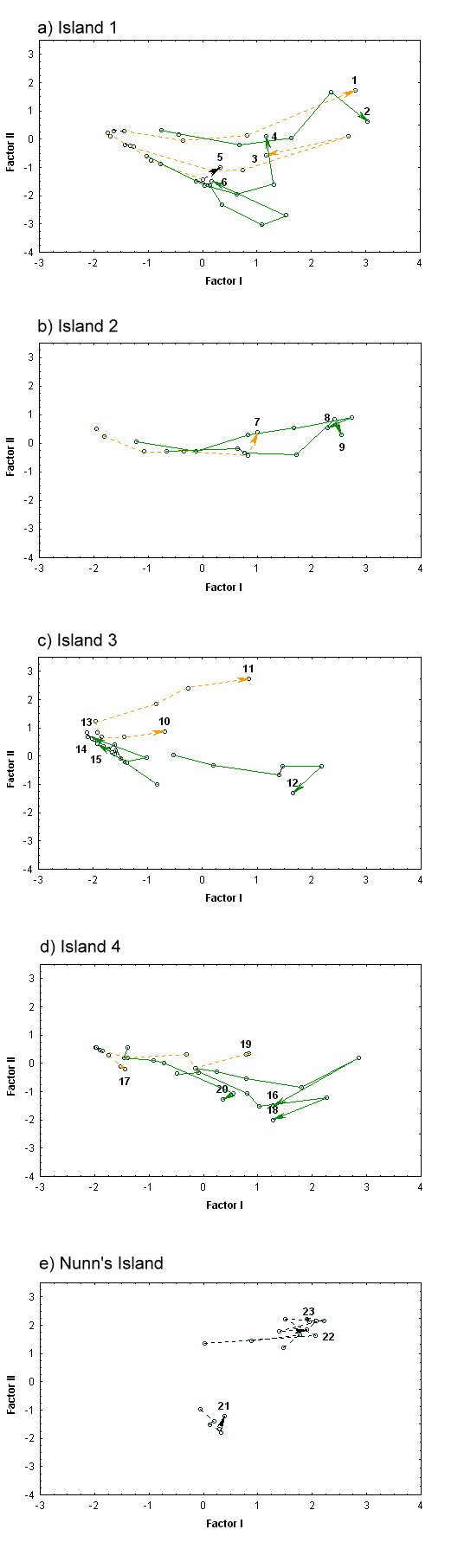
Vegetation dynamics were analyzed using principal components analysis (PCA), which allowed us to summarize and graph the changes in vegetation over time. Problems have been associated with PCA of highly variable data sets, but the method is effective where compositional gradients are less extreme (Gauch 1982), as is the case with intertidal marsh vegetation. The PCA was based on a species covariance matrix calculated from the mean cover class data for transects from
all sampling periods. Mean cover class was determined by summing the cover class values of species in each transect, and then dividing by the number of relevés examined. Mean cover across the transect is more suited for analyzing variation at the community scale, where the absence as well as occurrence of species is of interest. This allowed a comparison of the vegetation changes over time among transects on the separate islands. Interpretations of the first two PCA axes were derived from correlations of the factor scores with the transect means of species cover, species richness, total cover, and elevation. The elevation correlations were determined using the 1988 PCA transect scores, as this was the last year in which transects 10 and 11 were surveyed. The software program SYSTAT was used for all calculations.
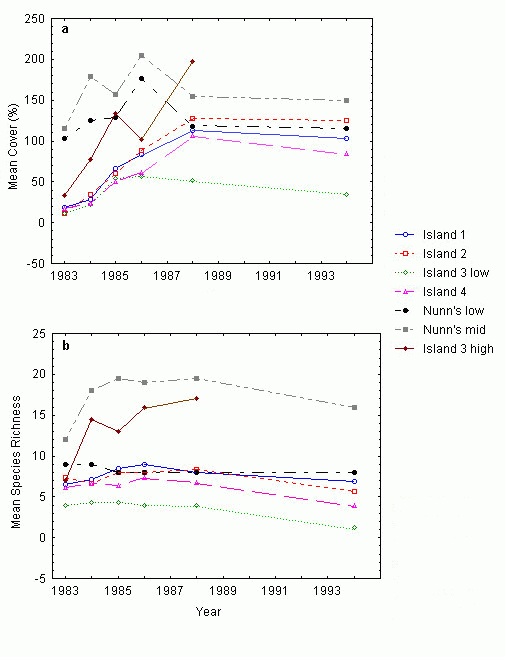
Changes in species composition and cover over time were also calculated separately for the various islands and treatments (i.e., planted blocks, unplanted blocks, and Nunn’s low- and mid-to-high reference marshes). Best estimates of mean cover-abundance for each species were calculated by summing the midpoints of each species’ Braun-Blanquet scale range (i.e., by setting r = 0.01,+= 0.5, 1 = 3, 2 = 15, 3 = 37.5, 4 = 67.5, 5 = 87.5) and dividing by the number of occurrences of the species within the data set. An estimate of total mean cover was then calculated by summing the mean cover values of each species within the data set. These data were used to compare mean cover between treatments and islands. The method tends to overestimate cover somewhat, but the results are relatively comparable. The frequency of occurrence of each species was calculated by dividing the number of occurrences of a species within the data set by the total number of relevés within the data set. Mean species richness values by treatment and by island were calculated by summing the total number of species in each transect and dividing by the number of transects in each treatment or island. We applied the cell-means ANOVA approach to the aboveground biomass data to test for differences between treatments and between islands.
During the autumn and winter of 1982, the islands in the estuary were subjected to high river discharges at low tides; however, there was no apparent damage or destabilization to the created islands (Brownlee et al. 1984). Continued observations to 1994 suggested that the islands remained stable, with no serious erosion occurring, even on island 3 directly adjacent to the main river channel.
Vegetation plug success
Plug success on the four constructed islands was 92% by July 1982. Most plug mortality occurred in the two low-elevation blocks of island 3 (transects 14 and 15). Plug mortality on island 3 increased from 13% in July 1982 to 28% in August 1983, and vegetation growth of the two low-elevation blocks never recovered. By August 1983, overall plug success on the four islands was about 90%. By 1984, it became too difficult to differentiate individual plugs among
the coalescing vegetation on many of the blocks (Fig. 3).